Magnesium has three naturally occurring isotopes with the following masses and natural abundances: Sketch the mass spectrum
Question:
Magnesium has three naturally occurring isotopes with the following masses and natural abundances: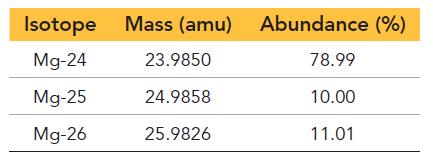
Sketch the mass spectrum of magnesium.
Transcribed Image Text:
Isotope Mg-24 Mg-25 Mg-26 Mass (amu) 23.9850 24.9858 25.9826 Abundance (%) 78.99 10.00 11.01
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
A mass spectrum is a graphical representation of the relative abundance of isotopes in ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Patients seeking care at the County General emergency room wait, on average, 6 minutes before seeing the triage nurse who spends, on average, 4 minutes assessing the severity of their problem. The...
-
An element has three naturally occurring isotopes with the following masses and abundances: Isotopic Mass (amu) Fractional Abundance 38.964 0.9326 39.964 1.000 104 40.962 0.0673 Calculate the atomic...
-
An element has three naturally occurring isotopes with the following masses and abundances: Isotopic Mass (amu) Fractional Abundance 27.977.. 0.9221 28.976.. 0.0470 29.974.. 0.0309 Calculate the...
-
Austin and Anya Gould are a middle-aged couple with two childrenRusty, age 13, and Sam, age 11whom they adopted this year. They also bought a new home in the area to give the children a yard in which...
-
Refer to the data for Balance, Inc., in Exercise 3-26. Required Using the Goal Seek function in Microsoft Excel, a. What number must Balance, Inc., sell to break even? b. What number must Balance,...
-
Calculate magnitude and location of the resultant force of water on this annular gate. 1m Water Gate 3 md --Hub-- 1.5m d P3.49
-
What is the System Production Phase, when does it start, and when does it end?
-
Dresser, Inc., manufactures three super-sports-hero dolls: Super Dunk, Pete Tulip, and Zonk. Production, however, is limited by the skilled labor necessary to produce these unique dolls. Data on each...
-
As discussed there are different interview styles that dictate how an investigator will seek information. One effective theme or style is for the investigator to sympathize with the suspect. In the...
-
Assume you have an graph as following. Use Dijkstra Algortihm to calculate shortest path from vertex A to every vertex (manuel calculation). Write the Dijkstra code to generate this graph and find...
-
The atomic mass of fluorine is 18.998 amu, and its mass spectrum shows a large peak at this mass. The atomic mass of chlorine is 35.45 amu, yet the mass spectrum of chlorine does not show a peak at...
-
Which pair of elements do you expect to be most similar? Why? a. Nitrogen and oxygen b. Titanium and gallium c. Lithium and sodium d. Germanium and arsenic e. Argon and bromine
-
Amelia Eyeland has been arrested for trespass, disorderly conduct, and resisting arrest. The charges stem from an incident in which Amelia and other members of the Green Warriors staged a raucous...
-
How do environmental factors, such as altitude, temperature, and atmospheric composition, influence respiratory physiology, necessitating adaptive responses at both the cellular and systemic levels...
-
How do the mechanisms of ventilation and gas exchange operate synergistically in facilitating the diffusion of oxygen and carbon dioxide across the respiratory membrane, ensuring optimal...
-
Can you elucidate the intricacies of cellular respiration, delineating the metabolic pathways involved and their respective roles in energy production within eukaryotic organisms ?
-
John Porter is an hourly employee of Motter Company located in New York City. This week, Porter had to travel to the company's regional office in Albany. He left Sunday at noon and arrived in Albany...
-
How do advancements in molecular biology, genomics, and systems biology deepen our understanding of the genetic determinants, molecular pathways, and regulatory networks governing respiratory...
-
Why are the seasons on Uranus different from the seasons on any other planet?
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
Using acetylene and 2-methylpropane as your only sources of carbon atoms, propose a plausible synthesis for 4-methyl-2-pentanone. You will need to utilize many reactions from previous chapters.
-
Draw a Lewis structure of a carbon atom that is missing one valence electron (and therefore bears a positive charge). Which second-row element does this carbon atom resemble in terms of the number of...
-
Below is the structure of caffeine, but its lone pairs are not shown. Identify the location of all lone pairs in this compound: - -N N. .C. Caffeine Z-O- Z-U
-
Counseling Services Goals: Observe activities in a school counseling office. The intern should interview both the mentor and at least one counselor about their respective roles in supporting student...
-
How does the allocation of resources (financial, human, and technological) influence organizational effectiveness, and what strategies can organizations use to optimize resource distribution ?
-
On September 1, 2025, Tamarisk Company sold 10,800 of its 10%, 15-year, $1,000 face value, nonconvertible bonds with detachable stock warrants at 102. Each bond carried three detachable warrants....

Study smarter with the SolutionInn App


