Name each alcohol. a. CH3-CH2-CH2 -OH CH2 -CH3 1 b. CH3 -CH-CH2 - 3 I OH
Question:
Name each alcohol.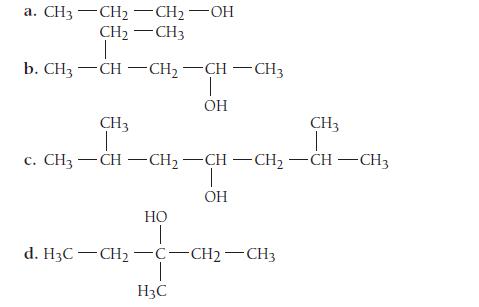
Transcribed Image Text:
a. CH3-CH2-CH2 -OH CH2 -CH3 1 b. CH3 -CH-CH2 -СН — СН3 I OH CH3 CH3 1 c. CH3-CH-CH2 -CH-CH2-CH-CH3 HO T OH T d. HzC-CH2 - С -СН2 — СН3 H3C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a 1propanol b ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
We already have the response for the journal entries but we need the others question then the ledger of the bank and embezzled (i'm talking about financial statement, income statement, and the ending...
-
Define the following terms Chemistry, Organic Chemistry, Ionic and Covalent bond, Isomer, Hydrocarbons, Functional group, Clinical Chemistry, Inductive effect How Friedrich Whler synthesize the first...
-
Name the phase transition in each of the following situations and indicate whether it is exothermic or endothermic: (a) Bromine vapor turns to bromine liquid as it is cooled. (b) Crystals of iodine...
-
A close company which prepares accounts to 31 March each year is owned and managed by a single shareholder/director who is not a Scottish taxpayer and who is paid a salary of 5,000 per month. In...
-
Under what conditions will the weighted average and FIFO methods give the same results?
-
Discuss how internal response moderators influence the customers response to the services-cape.
-
Consider the following cash flow profile, and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically...
-
The Sparrow Partnership plans to distribute $200,000 cash to its partners at the end of the year. Marjorie is a 40 percent partner and would receive $80,000. Her basis in the partnership is only...
-
asap please 1. Determine a maintenance priority by calculating Preventive Maintenance Factors (PMFs) for the three pieces of equipment listed in the table below. Clearly state which piece of...
-
Name each aldehyde or ketone. a. CH3 -C- CH - CH3 b. CH3CH,CH,CH,CH CH3 CH3 T C. CH3-C-CH-CH-CH-C-H CH3 d. CH3-CH-CH-C-CH3 CH CH3 ||
-
Draw the structure for each alcohol. a. 2-butanol b. 2-methyl-1-propanol c. 3-ethyl-1-hexanol d. 2-methyl-3-pentanol
-
You have determined that 2.1% of the CDs that your factory manufactures are defective due to a problem with materials and that 1.3% are defective due to human error. Assuming that these are...
-
Self-Study Problem 5.3 2022 AGI Phase-Out Ranges for Traditional and Roth IRA Contributions to be used for this problem. 2022 AGI Phase-Out Ranges for Deductible Traditional IRA Contributions Type of...
-
Using a spreadsheet program, construct a 30-year mortgage calculator and determine how long it takes to pay off the loan if a certain additional amount is paid towards principal each month. For...
-
Taxpayer: [Your Name] TIN: 987-654-321-000 Address: Poblacion, Puerto Galera, Oriental Mindoro On October 1, 2020, you have started and registered two (2) businesses: a bus transport company...
-
Starship plc specialises in the construction of retail shopping centres. This construction work is always based upon detailed customer specifications, and usually involves a fixed-price construction...
-
Calculate the Return on Equity: Netflix Income Statement Revenue Cost of Revenue Gross Profit Operating Expense Operating Income Int Exp Taxes Net Income From the Balance Sheet Shareholders Equity...
-
What are the critical components of financial management?
-
(a) Given a mean free path = 0.4 nm and a mean speed vav = 1.17 105 m/s for the current flow in copper at a temperature of 300 K, calculate the classical value for the resistivity of copper. (b)...
-
An AC current is given by I = 495 sin(9.43t), with I in mA and t in ms. Find (a) The rms current (b) The frequency in Hz.
-
What are the phase constants for the signals in Fig. 28.24? () (b) (c) ot 27 (e) (d) FIGURE 28.24 Exercise 17 Voltage
-
Find the rms current in a 1.0F capacitor connected across 120-V rms, 60-Hz AC power.
-
Accounts Receivable Analysis The following data are taken from the financial statements of Sigmon Inc. Terms of all sales are 2/10, n/45. 20Y3 20Y2 Accounts receivable, end of year Sales on account...
-
Required information Use the following information for the Exercises 3-7 below. (Algo) [The following information applies to the questions displayed below.] Laker Company reported the following...
-
III. Queen Spice Company is a business that sells high quality spices both online and in retail stores. The only warning label on Queen Spice Company's products is that the spice bottles should be...

Study smarter with the SolutionInn App


