Natural gas burns in air to form carbon dioxide and water, releasing heat. What minimum mass of
Question:
Natural gas burns in air to form carbon dioxide and water, releasing heat.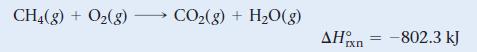
What minimum mass of CH4 is required to heat 55 g of water by 25 °C?
a) 0.115 g
b) 2.25 * 103 g
c) 115 g
d) 8.70 g
Transcribed Image Text:
CH4(g) + O2(g) CO2(g) + H2O(g) ΔΗ IX Π -802.3 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a...View the full answer

Answered By

Mercy Kangai
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past seven years. I can bring value to your business and help solve your administrative assistant issues. I have extensive experience in marketing and small business management.
4.80+
27+ Reviews
82+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
A gaseous hydrocarbon reacts completely with oxygen gas to form carbon dioxide and water vapor. Given the following data, determine Hof for the hydrocarbon: Hrxn = 2044.5 kJ/ mol Hof (CO2) = 393.5...
-
A 16.0-g sample of methane (CH 4 ) reacts with 64.0 g of oxygen gas in a container fitted with a piston (at 1.00 atm and 425 K). Methane can react with oxygen to form carbon dioxide and water vapor...
-
You are the Accounts Clerk for B2B Enterprises.You have been asked to complete the banking questions and bank reconciliation tasks below.To Do all tasks, the B2B EnterprisesOrganisationalPolicies and...
-
What arguments can be advanced in favor of treating fixed manufacturing overhead costs as period costs?
-
If v av = 0 for some time interval t , must the instantaneous velocity v be zero at some point in the interval? Support your answer by sketching a possible x -versus -t curve that has x = 0 for...
-
Do the following activities to complete your marketing plan: 1. Draw a simple organizational chart for your organization. 2. Develop a Gantt chart (see Chapter 2) to schedule the key activities...
-
Tampa Instrument Company manufactures gauges for construction machinery. The company has two production departments: Machining and Assembly. There are three service departments: Maintenance, Human...
-
Downsizing 1. In the trailer, Matt Damon and Kirsten Wigg are told that their $52,000 will be worth 12.5 million dollars in Leisure Land. Use a proportion to calculate how much $100,000 would be...
-
Explain how the sum of heat and work can be a state function, even though heat and work are themselves not state functions.
-
Which fuel is not a fossil fuel? a) Coal b) Hydrogen c) Natural gas d) Petroleum
-
The individual chromosomes become visible with a light microscope during which stage of mitosis? a. Prophase b. Prometaphase c. Metaphase d. Anaphase
-
In a big university, 70 % of the faculty members like to give plus and minus grades (such as B plus and C minus). The other 30 % of the faculty members do not like the plus and minus system. The...
-
What are some factors that affect a call options value?
-
Assume the tips received by five waitresses in a given weeknight are $25, $27, $28, $29, and $30. We draw two numbers randomly and take the average. Write the probability distribution of the sample...
-
Use a numerical integration routine on a graphing calculator to find the area in the first quadrant that is below the graph of And above the graph of y = 0.2x + 1.6. 6 = y 2 + 50 x
-
The newly produced 1992 Honda boasts 45 miles per gallon on the highway. Assume that the distribution of the miles per gallon is a normal distribution with a mean of 40 and a standard deviation of 5....
-
Situation In 2010, its first year of operations, Tara Corporation appropriately reported basic earnings per share of $1.05 on its income statement. During 2011 the company instituted a share option...
-
Refer to the table to answer the following questions. Year Nominal GDP (in billions) Total Federal Spending (in billions) Real GDP (in billions) Real Federal Spending (in billions) 2000 9,817 578...
-
Determine the bulk modulus for each of the following materials: (a) rubber, E r = 0.4 ksi, r = 0.48, and (b) glass, Eg = 8(10 3 ) ksi, g = 0.24.
-
The strain gage is placed on the surface of the steel boiler as shown. If it is 0.5 in. long, determine the pressure in the boiler when the gage elongates 0.2(10 -3 ) in. The boiler has a thickness...
-
The principal strains at a point on the aluminum fuselage of a jet aircraft are 1 = 780(10 -6 ) and 2 = 400(10 -6 ). Determine the associated principal stresses at the point in the same plane. E al...
-
Swathmore Clothing Corporation grants its customers 30 days' credit. The company uses the allowance method for its uncollectible accounts receivable. During the year, a monthly bad debt accrual is...
-
Company A has a sustainable ROE of 16% and a steady dividend payout ratio of 25%. What is the sustainable growth rate:
-
Carla Vista Company buys merchandise on account from Flint Company. The selling price of the goods is $840, and the cost of the goods is $480. Both companies use perpetual inventory systems....

Study smarter with the SolutionInn App


