Referring to Table 18.1, pick an indicator for use in the titration of each acid with a
Question:
Referring to Table 18.1, pick an indicator for use in the titration of each acid with a strong base.
a. HF
b. HCl
c. HCN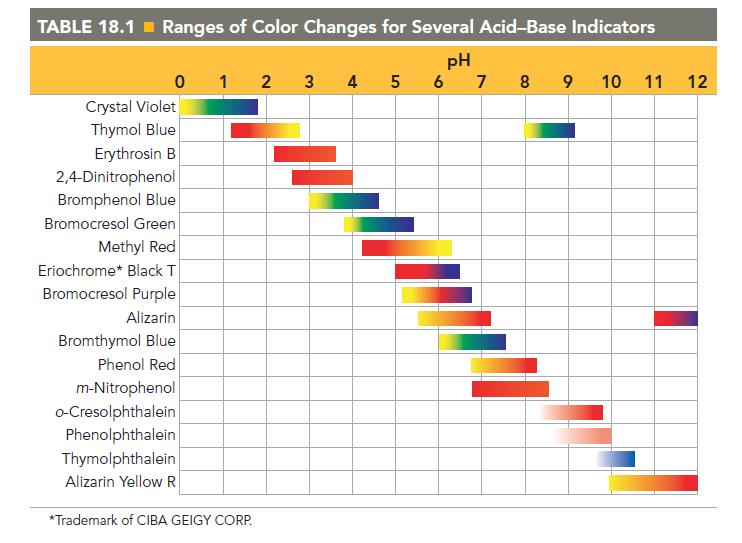
Transcribed Image Text:
TABLE 18.1 Ranges of Color Changes for Several Acid-Base Indicators pH 5 6 7 8 9 0 1 2 Crystal Violet Thymol Blue Erythrosin B 2,4-Dinitrophenol Bromphenol Blue Bromocresol Green Methyl Red Eriochrome* Black T Bromocresol Purple Alizarin Bromthymol Blue Phenol Red m-Nitrophenol o-Cresolphthalein Phenolphthalein Thymolphthalein Alizarin Yellow R *Trademark of CIBA GEIGY CORP. 3 4 tunnew 10 11 12
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a Phenol red mni...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Referring to Table 18.1, pick an indicator for use in the titration of each base with a strong acid. a. CH 3 NH 2 b. NaOH c. C 6 H 5 NH 2 TABLE 18.1 Ranges of Color Changes for Several Acid-Base...
-
Weak base B has a pK b of 6.78 and weak acid HA has a pK a of 5.12. a. Which is the stronger base, B or A ? b. Which is the stronger acid, HA or BH + ? c. Consider the following reaction: B(aq) +...
-
How does titration of a strong,monoprotic acid with a strong base differ from titration of a weak, monoprotic acid with a strong base with respect to the following: (a) Quantity of base required to...
-
Informative Speech Critique Evaluating Rhetorical Choices You may just number the questions and add a paragraph or two to answer - 1. Did you find the speaker credible? Why or why not? 2. Did the...
-
Bryant Bikes could sell its bicycles to retailers either assembled or unassembled. The cost of an unassembled bike is as follows. Direct materials .............. $150 Direct labor ............... 70...
-
Qualitative Characteristics SFAC No. 2 identifies the qualitative characteristics that make accounting information useful. Presented below are a number of questions related to these qualitative...
-
Is there a single standard command-line processor to parse and process argv?
-
One of the questions on a survey of 1000 adults asked if todays children will be better off than their parents (Rasmussen Reports website October 26, 2012). Representative data are shown in the file...
-
The table below shows the output per labourer per day for Guyana and Jamaica for Coffee and Rice. Coffee Rice Table 1 Output per labourer per day Guyana Jamaica 8 6 10 4 What is the autarky cost of...
-
A 20.0-mL sample of a 0.125 M diprotic acid (H 2 A) solution is titrated with 0.1019 M KOH. The acid ionization constants for the acid are K a1 = 5.2 * 10 -5 and K a2 = 3.4 * 10 -10 . At what added...
-
A 20.0-mL sample of 0.115 M sulfurous acid (H 2 SO 3 ) solution is titrated with 0.1014 M KOH. At what added volume of base solution does each equivalence point occur?
-
Howle Manufacturing Company began operations on January 1. During the year, it started and completed 1,700 units of product. The company incurred the following costs. 1. Raw materials purchased and...
-
Explain why German reparation payments were essential to the survival of the international financial system in the 1920s
-
Explain the concept of inflation in your own terms. How have you been impacted by inflation in your lifetime? What did you do to offset the rising costs?
-
Elucidate the interconnection between labeling theory and symbolic interaction theory, highlighting their conceptual synergy. Additionally, furnish an illustrative example demonstrating the...
-
Fanblade, Inc. is a manufacturer of fan blades for jet engines. These blades are where cool air ranging from -50oC to +50oC enters the jet engine. The fan blades are exposed to foreign objects such...
-
Zoe (Gemma's replacement - how quickly is Gemma replaced? Brutal!) raises concerns with the partner, Will, about: irregularities with the purchase of artwork the tax issue irregularities with the...
-
When selecting short burst quotations for your Formal Research Assignment, what criteria will you consider? What makes for a strong quotation? How long can a short burst quotation be?
-
What can scientists learn by comparing the fossilized skeletons of extinct primates with the bones of modern species?
-
To fill in some of the details in the derivation of the smallest phase increment separating two resolvable FabryPerot fringes, that is, satisfy yourself that Show that Eq. (9.72) can be rewritten as...
-
Consider the interference pattern of the Michelson Interferometer as arising from two beams of equal flux density. Using Eq. (9.17), compute the half-width. What is the separation, in δ,...
-
Satisfy yourself of the fact that a film of thickness /4 and index n 1 will always reduce the reflectance of the substrate on which it is deposited, as long as n s > n 1 > n 0 . Consider the...
-
Which solution did the Farmers' Alliance movement of the late nineteenth century initially propose to solve farmers' economic problems?
-
Identify the statements that apply to CHAMPVA's cost to patients. Multiple select question. In most cases, CHAMPVA pays equivalent to Medicare/TRICARE rates. Beneficiaries are not responsible for...
-
Discuss a means used by the World Bank or the World Trade Organization that might assist the developing country in removing barriers to contributing to the global economy

Study smarter with the SolutionInn App


