The amount of strong acid in the flask shown here is to be titrated by a strong
Question:
The amount of strong acid in the flask shown here is to be titrated by a strong base. Which mark on the burette next to the flask indicates the amount of base required to reach the equivalence point?
(a) A
(b) B
(c) C
(d) D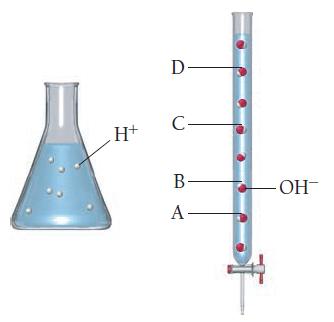
Transcribed Image Text:
H+ D- C- B- A- -ОН-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
d Because the flask ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
ABCD is a square with side length 2. Quantity A is greater. Quantity B is greater. The two quantities are equal. The relationship cannot be determined from the information given. B A C D
-
A small sphere of weight W is held as shown by two wires AB and CD. Ifwire AB is cut, determine the tension in the other wire(a) Before AB is cut,(b) Immediately after AB has beencut. A 50 B 70 D
-
Identify the reagents necessary to achieve each transformation below: (a) (b) (c) D D D
-
Google, the immensely popular Web search engine, has been touted as the closest thing the Web has to an ultimate answer machine. Although this is debatable, of course, it is far more difficult to...
-
Each day, Adama Corporation processes 1 ton of a secret raw material into two resulting products, AB1 and XY1. When it processes 1 ton of the raw material, the company incurs joint processing costs...
-
The financial statements of Marks and Spencer plc (M&S) are available at the books companion website or can be accessed at http:/corporate.marksandspencer.com/...
-
Steven Sanchez worked as a warehouse employee for Gruma Corporation. In December 2016, Sanchez was presented an arbitration agreement during a meeting with George Tate, the human resources manager...
-
Accounting for Patents, Franchises, and R&D Devon Harris Company has provided information on intangible assets as follows. A patent was purchased from Bradtke Company for $2,500,000 on January 1,...
-
Assume a simultaneous open market purchase of 100 million from the Bank of England and a repayment of a discount loan of 5 million from Bank A to the Bank of England. Show the overall change in their...
-
A 50.0-mL sample of 0.200 M sodium hydroxide is titrated with 0.200 M nitric acid. Calculate pH: (a) After adding 30.00 mL of HNO 3 (b) At the equivalence point
-
A 1.0-L buffer solution is 0.10 M in HF and 0.050 M in NaF. Which action destroys the buffer? (a) Adding 0.050 mol of HCl (b) Adding 0.050 mol of NaOH (c) Adding 0.050 mol of NaF (d) None of the above
-
Bob and Sue were expecting a baby in January, but Sue was rushed to the hospital in December. She delivered the baby but it died the first night. Are Bob and Sue entitled to a dependency exemption...
-
How can a company deal with strategic exposure?
-
What cultural issues would you anticipate in a planned partnership between a UK listed e-company and a Korean family conglomerate?
-
Francisco takes out a student loan from the First National Bank. After graduation, Francisco goes to work, but he does not make payments on the loan. The bank agrees with Ace Collection Agency that...
-
What does it mean to take an instrument in good faith?
-
If a bank pays a stale check in good faith without consulting the customer, the bank cannot charge the customers account. (TRUE/FALSE)
-
What variables might be used to segment these industrial markets? (a) Industrial sweepers, (b) Photocopiers, (c) Computerized production control systems, (d) Car rental agencies.
-
Consider the reaction of acetic acid in water CH 3 CO 2 H(aq) + H 2 O(l) CH3CO 22 (aq) + H 3 O + (aq) where Ka 5 1.8 3 1025. a. Which two bases are competing for the proton? b. Which is the stronger...
-
The load resistance R L in Fig. 11.51 is adjusted until it absorbs the maximum average power. Calculate the value of R L and the maximum average power. > 120 .-j10 40
-
The variable resistor R in the circuit of Fig. 11.50 is adjusted until it absorbs the maximum average power. Find R and the maximum average power absorbed. -j100 2 100 2 100 2 100/0 V (+ j100 2 lle
-
Find the value of Z L in the circuit of Fig. 11.49 for maximum power transfer. 60/0 V 40 2 -j10 2 40 2 80 2 5/0 A ZL j40 2
-
Down Under Products' sales budget for the next four months is as follows: Unit Sales April 80,000 May June July 90,000 120,000 95,000 The company wants its ending Inventory levels to equal 10% of the...
-
Recording Entries for Finance Lease with Unguaranteed Residual Value-Lessee Lessor Corporation, a manufacturer of equipment, enters into a lease of specialized equipment with Lessee Corp. on January...
-
? Weighted average cost method with perpetual inventoryWeighted average cost method with perpetual inventoryExhibit 5 ?Entries and Perpetual Inventory Account ( Weighted Average ) The beginning...

Study smarter with the SolutionInn App


