The first diagram depicts a compound in its liquid state. Which of the other diagrams best depicts
Question:
The first diagram depicts a compound in its liquid state.
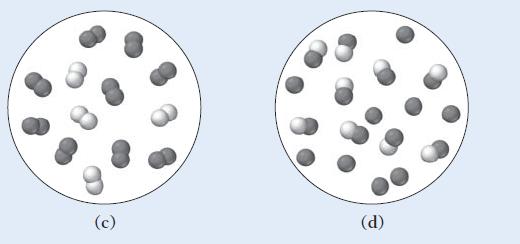
Which of the other diagrams best depicts the compound after it has evaporated into a gas?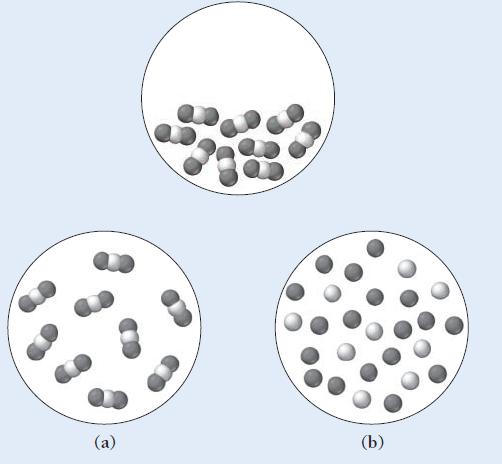
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)

Answered By

Mary Boke
As an online tutor with over seven years of experience and a PhD in Education, I have had the opportunity to work with a wide range of students from diverse backgrounds. My experience in education has allowed me to develop a deep understanding of how students learn and the various approaches that can be used to facilitate their learning. I believe in creating a positive and inclusive learning environment that encourages students to ask questions and engage with the material. I work closely with my students to understand their individual learning styles, strengths, and challenges to tailor my approach accordingly. I also place a strong emphasis on building strong relationships with my students, which fosters trust and creates a supportive learning environment. Overall, my goal as an online tutor is to help students achieve their academic goals and develop a lifelong love of learning. I believe that education is a transformative experience that has the power to change lives, and I am committed to helping my students realize their full potential.
5.00+
4+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Use implicit differentiation to find dy/dx. 6x 3 + 7y 3 = 13xy
-
William Bisby gave an all-terrain vehicle (ATV) to Del City Cycle in Enid, Oklahoma, to sell on his behalf. Joseph Maddox bought the ATV but paid for it with a check written on a closed checking...
-
Your division is considering two investment projects, each of which requires an up-front expenditure of $25 million. You estimate that the cost of capital is 10 percent and that the investments will...
-
T. Christian Cooper was a partner to Sanders and Richard Campbell d/b/a The Mullen Company. In 2001, Cooper helped bring about a management agreement between The Mullen Co. and Newnan Crossing...
-
Union Bay Plastics is investigating the purchase of automated equipment that would save $100,000 each year in direct labor and inventory carrying costs. This equipment costs $750,000 and is expected...
-
Car Crash Investigation Background information: A collision occurred involving two vehicles on Route 28 N. The speed limit in this zone is 45 mph. A 2011 Honda Odyssey minivan was stopped at the...
-
At December 31, 2019, Catu Medical reported the following information on its statement of financial position. Accounts receivable R$960,000 Less: Allowance for doubtful accounts 80,000 During 2020,...
-
Find the radius (r) in centimeters of a spherical water droplet with a volume (V ) of 0.058 cm 3 . For a sphere, V = (4>3) r 3 . GIVEN: V = 0.058 cm FIND: r in cm CONCEPTUAL PLAN V 4 v=mr V= 3 r...
-
Find the density (in g/cm 3 ) of a metal cylinder with a mass (m) of 8.3 g, a length (l ) of 1.94 cm, and a radius (r) of 0.55 cm. For a cylinder, V = r 2 l. GIVEN: m = 8.3 g 1, r FIND: d in g/cm...
-
What type of layout(s) would be appropriate for: a. A grocery store? b. Home construction? c. Electronics assembly? d. A university?
-
What is a good problem statement for high turnover rates in fast food?
-
Issuing Common Stock Valdez Manufacturing sold 22,400 shares of common stock to stockholders at the time of its incorporation. Valdez received $24.40 per share for the stock. Required: Question...
-
A horizontal rectangular plate, simply supported on all four edges, with dimensions 1.3 m and 1.3 m , with a thickness 6 mm is subjected to a vertically distributed load of 5000 N/m2. Knowing that...
-
The enthalpy of vaporization of water is 2255 J/g. Assume AHvap to be independent of temperature and pressure and calculate the vapor pressure of water at 80 C.
-
Identify Google's Mission and its key values Describe Google's Culture. why this culture is as it is, what it is trying to obtain/achieve? Give at least two examples of cultural artifacts and/or...
-
Prospect Realty Co. pays weekly salaries of $27,600 on Monday for a six-day workweek ending the preceding Saturday. Journalize the necessary adjusting entry at the end of the accounting period,...
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
Consider the reaction: CO (g) + 2 H 2 (g) CH 3 OH (g) A reaction mixture initially contains [CO] = 0.600 M and [H 2 ] = 1.20 M. At equilibrium, the CO concentration is found to be 0.100 M. Find the...
-
Consider the reaction: 2 COF 2 (g) CO 2 (g) + CF 4 (g) Kc = 2.00 In an equilibrium mixture, the concentration of COF 2 is 0.35 M and the concentration of CO 2 is 0.144 M. What is the equilibrium...
-
Consider the reaction: N 2 O 4 (g) 2 NO 2 (g) Kc = 0.36 A reaction mixture initially contains [N 2 O 4 ] = 0.100 M. Find the equilibrium concentrations of N 2 O 4 and NO 2 .
-
John, an executive at Company X, uses his company expense account for his health club membership. This is an example of Question 5 options: whistleblowing insider trading abuse of official position...
-
Cobra, Incorporated, a sporting goods retailer, estimates to sell 50,000 units in the next quarter. The beginning inventory is 4,000 units and ending inventory is estimated to be 20 percent of...
-
What is a hostel? Select one: It is a home made with natural materials obtained from the environment. It is a place that offers refuge to pilgrims or care to vulnerable groups. It is a maritime or...

Study smarter with the SolutionInn App


