The tabulated data show the rate constant of a reaction measured at several different temperatures. Use an
Question:
The tabulated data show the rate constant of a reaction measured at several different temperatures. Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction.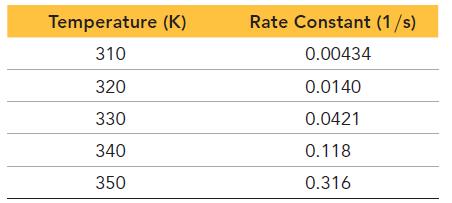
Transcribed Image Text:
Temperature (K) 310 320 330 340 350 Rate Constant (1/s) 0.00434 0.0140 0.0421 0.118 0.316
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The Arrhenius equation relates the rate constant k the activation energy E the gas constant R and th...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The tabulated data show the rate constant of a reaction measured at several different temperatures. Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction....
-
The following data show the rate constant of a reaction measuredat several different temperatures. Temperature (K) Rate Constant (1/s) 300 7.5610 - 2 310 0.221 320 0.605 330 1.56 340 3.79 Part A Use...
-
The data shown here were collected for the first-order reaction: Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction. NO(g) N(g) + O(g)
-
Like Death Valley, the Dead Sea is a place where you can walk on dry land below sea level, and an apparently unmotivated hole in the ground along a strike-slip fault. How do you think it formed?
-
Dalton Inc. produces and sells three products. Unit data concerning each product is shown below. The company has 2,000 hours of labor available to build inventory in anticipation of the company's...
-
Solve this system of two equations with two unknowns EES: x3 y2 = 7.75 3xy + y2 = 3.5
-
Define tolerable error.
-
1. Refer to Exhibit 1-8, which lists factors relevant for choosing an overseas market for listing or raising capital. Which factors might have been relevant in E-centives decision to raise capital...
-
Discuss the role of kernel-level synchronization primitives, such as spinlocks, semaphores, and mutexes, in ensuring safe concurrent access to shared resources. How does improper synchronization lead...
-
A reaction has a rate constant of 0.000122/s at 27 C and 0.228/s at 77 C. a. Determine the activation barrier for the reaction. b. What is the value of the rate constant at 17 C?
-
The tabulated data were collected for the second-order reaction: Cl(g) + H 2 (g) HCl(g) + H(g) Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction....
-
Vairs Steel Parts produces parts for the automobile industry. The company has monthly fixed expenses of $600,000 and a contribution margin of 80% of revenues. Requirements 1. Compute Vairs Steel...
-
Emma owns a small artisanal chocolate business. She sells her handcrafted chocolates at local markets and supplies them to a few independent boutique stores in her town. For the chocolates sold at...
-
Read the facts on the Marina Hotel and Fourth of July Detour cases and respond to the questions following: 1. Marina Hotel: On or about November 22, Margot was attending a conference by El Mundo...
-
A college student realized that he was spending too much money on movies. For the remaining 5 months of the year his goal is to spend a mean of $40 a month towards movies. How much can he spend in...
-
Given the facts of the case, the legal issues I raised and the brief answers how would I "word" or "sum" the legal analysis based on the summary of both cases into the memo? INTRODUCTION You have...
-
Mr. Morani doesn't fall under the provisions of A112(2), or A112(3), and is eligible to apply for PRRA. Mr. Morani has no previous refugee or PRRA claims, so he could present all the personal and...
-
McKnight, Inc. manufactures bookcases and uses an activity- based costing system. McKnights activity areas and related data follow: McKnight produced two styles of bookcases in April: the standard...
-
Find the center of mass of a thin triangular plate bounded by the y-axis and the lines y = x and y = 2 - x if (x, y) = 6x + 3y + 3.
-
Find i 0 (t) for t > 0 in the circuit in Fig. 16.72 . 2 + Vo 1 7.5e-2t u(t) V ( +) 4.5[1 u(t)]V 0.5v. 1H
-
Determine i o (t) in the network shown in Fig. 16.70 . 20 + 40u(t) V (t 2 H -/4
-
Determine i o (t) in the circuit in Fig. 16.69 . 2 H ele 5e-2'u(t) A 12
-
Mark Wellaby, MD, practices medicine. During the year he received the following items in payment for his services: cash of $48,000; farm produce worth $1,000; common stock with a total par value of...
-
The following data reports on the July production activities of the Molding department at Ash Company. Beginning work in process Direct materials Conversion Costs added this period. Direct materials...
-
Discuss the various query languages used in databases, such as SQL, MDX, and CQL. Explain the differences and when each language is used. Provide examples of queries written in different languages.

Study smarter with the SolutionInn App


