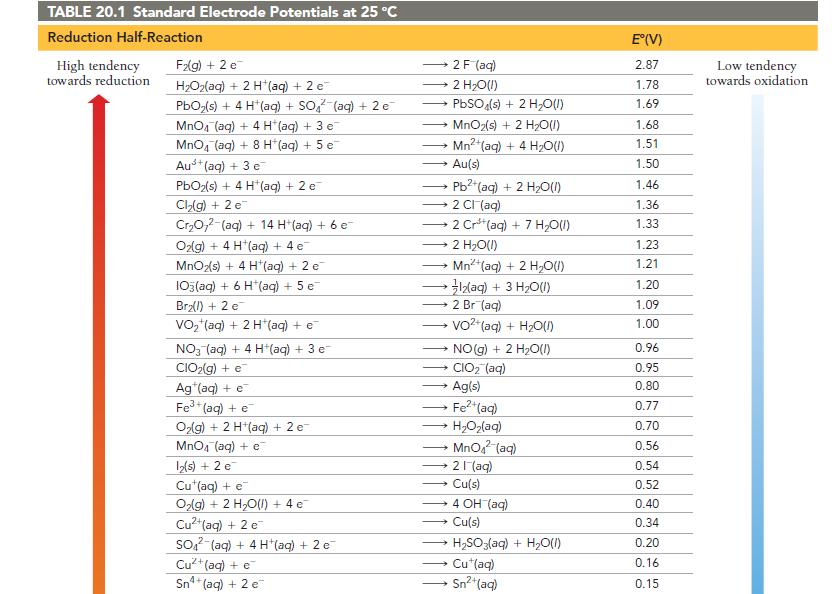
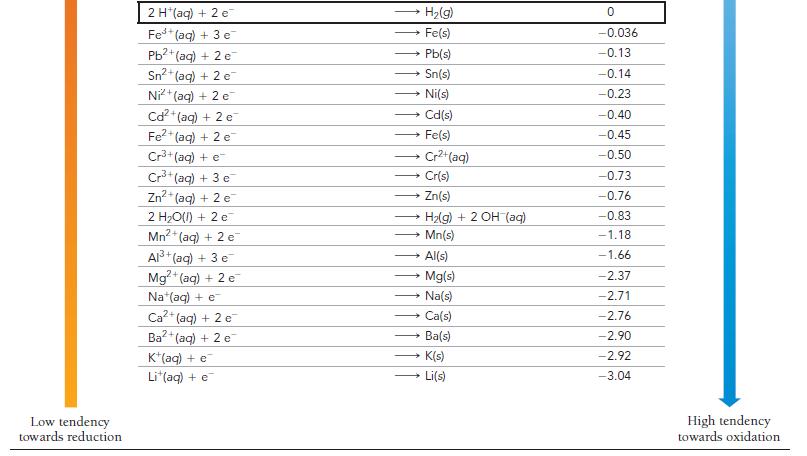
Use Table 20.1 to calculate G for the reaction. 2 MnO4 (aq) + Cd(s) - a) +30.9
Question:
Use Table 20.1 to calculate ΔG° for the reaction.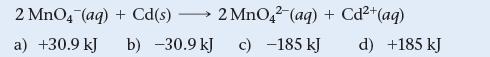
Transcribed Image Text:
2 MnO4 (aq) + Cd(s) - a) +30.9 kJ 2 MnO42 (aq) + Cd²+ b) -30.9 kJ c) -185 kJ (aq) d) +185 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
c...View the full answer

Answered By

Amar Kumar Behera
I am an expert in science and technology. I provide dedicated guidance and help in understanding key concepts in various fields such as mechanical engineering, industrial engineering, electronics, computer science, physics and maths. I will help you clarify your doubts and explain ideas and concepts that are otherwise difficult to follow. I also provide proof reading services. I hold a number of degrees in engineering from top 10 universities of the US and Europe.
My experience spans 20 years in academia and industry. I have worked for top blue chip companies.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Carbon disulfide (CS2) is a toxic, highly flammable substance. The following thermodynamic data are available for CS2(l) and CS2(g) at 298 K: (a) Draw the Lewis structure of the molecule. What do you...
-
GSSG + NADPH + H+ 2GSH + NADP+ (a) Calculate G for the glutathione reductase reaction in the direction shown, using E values from Table 14.1. (b) Suppose that a cell contained an isoform of...
-
Using data from Appendix C, calculate G for the following reactions. Indicate whether each reaction is spontaneous at 298 K under standard conditions. 2 SO2(g) + O2(g) 2 SO3(g) NO2(g) + N20(g)-3N0(g)...
-
Solve the inequality. Write the solution in interval notation. |15 = x < 7
-
Books-For-All is a well-established chain of 20 bookstores in western Ohio. In recent years, the company has grown rapidly, adding five new stores in regional malls. The manger of each store selects...
-
Seguin Inc. has the following projected unit sales for the first four months of 2011: January .........102,400 February ....... 96,000 March ........128,000 April ..........153,600 Company policy is...
-
The Nielsen family formed their corporation, N. Robert Nielsen, Inc., to conduct farming operations. Morre, Grider & Co. is a certified public accounting firm that has provided accounting, tax, and...
-
Miyamoto Jewelers is considering a special order for 10 handcrafted gold bracelets to be given as gifts to members of a wedding party. The normal selling price of a gold bracelet is $389.95 and its...
-
What output will be produced by the following code segment? int divide (double a, double b}{ } return (a/b); int main(){ double x=1.0, y = 4.0; cout < < divide(x, y); return 0; }
-
Consider the reaction at 298 K: Calculate G rxn under these conditions: Is the reaction more or less spontaneous under these conditions than under standard conditions? 2 NO(g) + O(8) O(g) 2 NO(8)...
-
Arrange these gases in order of increasing standard molar entropy: SO 3 , Kr, Cl 2 . (a) Kr < Cl < SO3 (c) SO3 Cl < Kr (b) Kr < SO3 < Cl (d) Cl < Kr < SO3
-
Refer to the information in QS 9-5 and prepare the journal entry assuming the note is honored by the customer on October 31, 2011.
-
You work for a nuclear research laboratory that is contemplating leasing a diagnostic scanner (leasing is a very common practice with expensive, high-tech equipment). The scanner costs $6,500,000 and...
-
You find a zero coupon bond with a par value of $10,000 and 21 years to maturity. The yield to maturity on this bond is 4.3 percent. Assume semiannual compounding periods. What is the price of the...
-
your bank pay 5 percent annual interest compounded semianually on your savings account. you dont expect to add to the current balance of $2,700 over the next four year. how much money can you expect...
-
You are building a view that includes Cost and Sale Price data. When you add the Cost field to the view, a prefix of $ is included. When you add the Sale Price field to the view, no prefix is...
-
KK company is your supplier. You need to pay the company for some inventories that are worth $100,000. KK company offers you a credit terms of 1/10 net 70. Compute the implied interest to you if you...
-
Electrolex, Inc., has four-year bonds outstanding that pay a coupon rate of 8.83 percent semiannually. If these bonds are currently selling at $861.63, the yield to maturity that an investor can...
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Coaxial cables are widely used with audio-visual technology, electronic instrumentation, and radio broadcasting, because they minimize interference with or from signals traveling on the cable....
-
A thin rod of length L carries charge Q distributed uniformly over its length. (a) Show that the potential in the plane that perpendicularly bisects the rod is given by where r is the perpendicular...
-
Two closely spaced square conducting plates measure 10 cm on a side. The electric-field energy density between them is 4.5 kJ/m 3 . Whats the charge on the plates?
-
Three resistors are connected in series in a circuit that is powered by a battery, and the individual resistance values are 250 Ohms, 150 Ohms and 50 Ohms. The current flowing in the circuit is 0.05...
-
6.) In the figure, find the value of the unknown resistor R if I = 6A and AV = 6V. R ww av on isa 10 R =
-
Consider a charge q being injected into an area with a homogeneous electric field as shown: V Y Co q Z X Complete the following statements: The electric field will The x-component of the charge's...

Study smarter with the SolutionInn App


