Water (a) wets some surfaces and beads up on others. Mercury (b), in contrast, beads up on
Question:
Water (a) “wets” some surfaces and beads up on others. Mercury (b), in contrast, beads up on almost all surfaces. Explain this difference.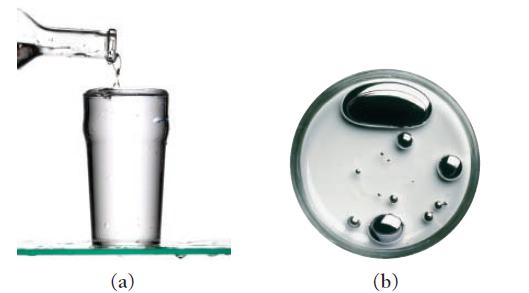
Transcribed Image Text:
1 (a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
The difference in wetting behavior between water and mercury is primarily due to the relative streng...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Reprinted with permission from The Parable of the Sadhu, by Bowen H. McCoy, Harvard Business Review. Copyright Harvard Business Publishing. Last year, as the first participant in the new six-month...
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
State whether the following statements are true or false. Also, very briefly mention the reason if the statement is false. i. TOFO Surfactants increase the surface tension of water. ii. TOFO The Mach...
-
Sam Simpleton, a resident of Kansas, and Nellie Naive, a resident of Missouri, each bought $85,000 in stock at local offices in their home States from Evil Stockbrokers, Inc. (Evil), a business...
-
Under what circumstances might the IRR and NPV approaches have conflicting results?
-
What ethical problems should you be aware of in searching social medial sites?
-
Aaron, a resident of Minnesota, has been a driver for Green Delivery Service for the past six years. For this purpose, he leases a truck from Green, and his compensation is based on a percentage of...
-
Deep Freeze produces fleece jackets. The company uses JIT costing for its JIT production system. Deep Freeze has two inventory accounts: Raw and In-Process Inventory and Finished Goods Inventory. On...
-
The structures of two isomers of heptane are shown. Which of these two compounds would you expect to have the greater viscosity? Compound A Compound B
-
Which compound would you expect to have greater surface tension: acetone [(CH 3 ) 2 CO] or water (H 2 O)? Explain.
-
Is it harder to find statistical significance using a test with level 0.05 or a test with level 0.01? In other words, would a test that is statistically significant using 0.05 always be statistically...
-
What are the causes of death that increase during a recession?
-
Analyze the specific anti-money laundering (AML) and counter-terrorist financing (CTF) regulations that commercial banks must adhere to. Explain the measures and procedures in place for customer due...
-
Your rich uncle has promised to give you $2,000 one year from today. After that, your uncle will increase the payout by 2% every year in perpetuity. Assuming a discount rate of 5% p.a., how much is...
-
1. Suppose that the telephone calls arriving at a particular switchboard follow a Poisson process with an average of 10 calls coming per hour. Find each of the following, and round your answers to...
-
. Below is the table summarizing the PVT properties of CO(g) (n=1 mol) at 313K. Calculate the products of PV at the given pressures using the Van der Waals EOS. P(atm) PV(obs.) (atm L/g) PV(calc.)...
-
Of its monthly sales, The Kingsman Company historically has had 25-percent cash sales with the remainder paid within one month. Each months purchases are equal to 75 percent of the next months sales...
-
For the following exercises, find the inverse of the function and graph both the function and its inverse. f(x) = 4 x 2 , x 0
-
Draw the resonance structures of a fully deprotonated phosphatidic acid.
-
Octanol is more efficient than hexanol at crossing the cell membrane and entering a cell. Explain.
-
Would you expect glycerol to readily cross the membrane?
-
Describe the collaborative roles of the team leader and the team coach in helping a group of people come together to form a team. Recommend strategies for Alex as team leader to use in helping to...
-
Although this talk is mostly about the personal finance crisis impacting older generations and Baby Boomers, what about the personal finance crisis impacting younger people like yourself? Why do you...
-
How much work is required to move a + 1 . 0 0 C charge from a point at an electric potential of + 4 0 0 . V to another at + 5 5 0 . V ?

Study smarter with the SolutionInn App


