What is the molarity of a 6.56% by mass glucose (C 6 H 12 O 6 )
Question:
What is the molarity of a 6.56% by mass glucose (C6H12O6) solution? (The density of the solution is 1.03 g/mL.)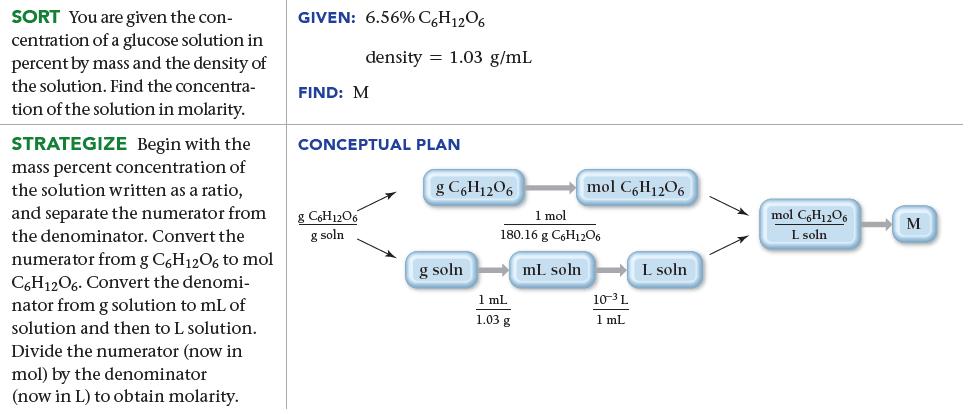
Transcribed Image Text:
SORT You are given the con- centration of a glucose solution in percent by mass and the density of the solution. Find the concentra- tion of the solution in molarity. STRATEGIZE Begin with the mass percent concentration of the solution written as a ratio, and separate the numerator from the denominator. Convert the numerator from g C6H12O6 to mol C6H12O6. Convert the denomi- nator from g solution to mL of solution and then to L solution. Divide the numerator (now in mol) by the denominator (now in L) to obtain molarity. GIVEN: 6.56% C6H12O6 density 1.03 g/mL FIND: M CONCEPTUAL PLAN 8 C6H12O6 g soln g C6H12O6 g soln mol C6H1206 1 mol 180.16 g C6H1206 ml. soln 1 mL 1.03 g 10-³L 1 mL L soln mol C6H1206 L soln M
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
656 g C6H1206 x 100 g soln X 1 m...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
What is the molarity of a solution made when 66.2 g of C6H12O6 are dissolved to make 235 mL of solution?
-
What is the molarity of a solution made by dissolving 332 g of C6H12O6 in 4.66 L of solution?
-
Show the parse trees for the two parses that the grammar assigns for sentence S1. S1: the train station bus rumbles [3 marks] (b) Give an algorithm for a bottom-up passive chart parser without...
-
What are the three viewpoints of product life cycle? How do they differ?
-
The following is a list of costs that were incurred in the production and sale of boats: a. Commissions to sales representatives, based upon the number of boats sold. b. Cost of boat for grand prize...
-
Donna White started her practice as a design consultant on January 1, 2010. During the first month of operations, the business completed the following transactions: Requirements 1. Open the following...
-
From its first Red Lobster in 1968, Darden Restaurants has grown the chain to 690 locations, with over $2.6 billion in U.S sales annually. The casual dinning market may be crowded, with competitors...
-
Exhibit 4 - Resource Allocation Happiland is dealing with some capacity challenges in their candy division. They currently produce three types of sour candies, Sugarbombs, Cherrysqueezes, and...
-
A solution contains 22.4 g glucose (C 6 H 12 O 6 ) dissolved in 0.500 L of water. What is the molality of the solution? (Assume a density of 1.00 g/mL for water.) a) 0.238 m b) 44.8 m c) 0.249 m d)...
-
A solution is prepared by dissolving 17.2 g of ethylene glycol (C 2 H 6 O 2 ) in 0.500 kg of water. The final volume of the solution is 515 mL. Calculate the concentration of the solution in each...
-
The chief executive officer of Acadia, Inc. attended a conference in which one of the sessions was devoted to variable costing. The CEO was impressed by the presentation and has asked that the...
-
Part 1Sales related and purchase related transactions. The following were selected from among the transactions completed by the Brown Company duringMay of the current year: Prepare journal entries...
-
Do you think the must-carry rules violate a cable company's First Amendment rights? Why or why not?
-
. A qualitative study of racial minority single mothers' work experiences. https://psycnet.apa.orq/record/2018 -49845-001 annotated bibliography Article One: List all important reference information...
-
As the appointed strategist for a South African apparel retail store chain, you have observed a shift in customer preferences over the past two years. What was once favoured by customers is no longer...
-
Suppose a pay-as-you-go social security system where social security is funded by a proportional tax on the income of the young. That is, the tax collected by the government is yy, where y is the tax...
-
Does a corporation with a positive balance in Retained Earnings have an equivalent amount of cash on hand? Explain.
-
Using a graphing utility, graph y = cot -1 x.
-
Oil with a specific gravity of 0.90 is flowing downward through the venturi meter shown in Fig. 6.33. If the velocity of flow in the 2-in-diameter section is 10.0 ft/s, calculate the deflection h of...
-
Oil with a specific gravity of 0.90 is flowing downward through the venturi meter shown in Fig. 6.33. If the manometer deflection h is 28 in, calculate the volume flow rate of oil. 4-in inside...
-
The venturi meter shown in Fig. 6.32 carries oil (sg = 0.90). The specific gravity of the gage fluid in the manometer is 1.40. Calculate the volume flow rate of oil. 75-mm inside diameter Flow 0.25 m...
-
A patient is administered 1 3 1 . How long will it take for observed radioactivity in her body to decrease to one - fourth of its original magnitude? If the half-life of the 1 3 1 is 8 . 1 days
-
Joseph borrowed $1800 from the bank at 7.6% per annum calculated on the unpaid monthly balance. He agreed to repay the loan in blended payments of $220 per month. How much of the first payment is...
-
A pioneering e-commerce platform is helping women in East Africa access menstrual care, personal care and women's health products. The platform is optimised to provide confidential access to products...

Study smarter with the SolutionInn App


