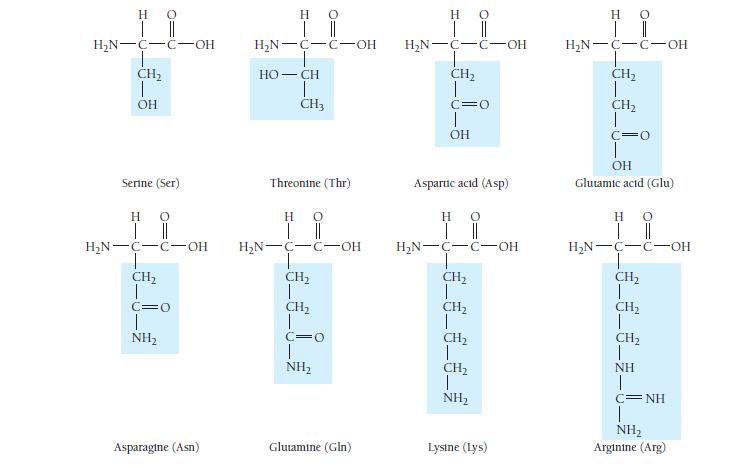
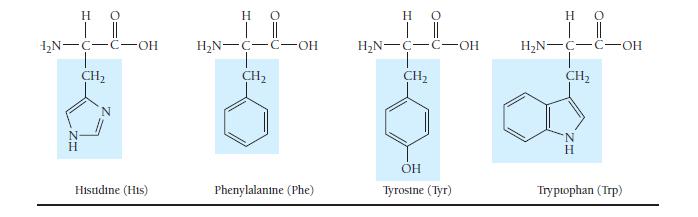
Which amino acids in Table 23.3 are most likely to be involved in hydrophobic interactions? TABLE 23.3
Question:
Which amino acids in Table 23.3 are most likely to be involved in hydrophobic interactions?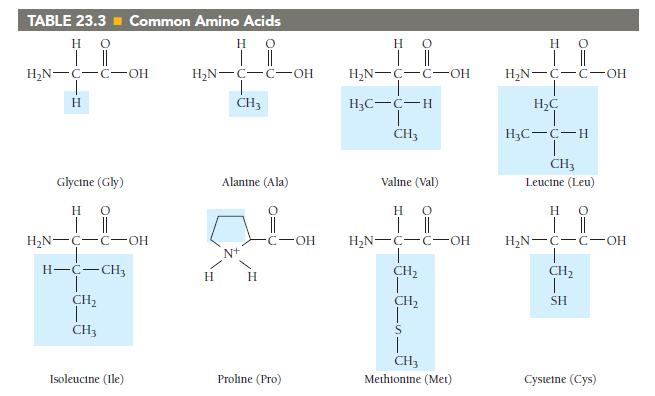
Transcribed Image Text:
TABLE 23.3 Common Amino Acids Η Ο || | Η Ο || | H₂N-C-C-OH H Glycine (Gly) HO | || H₂N-C-C-OH H-C-CH3 CH₂ CH3 Isoleucine (Ile) H₂N-C-C-OH T CH3 H Alanine (Ala) H C-OH Proline (Pro) Η | H₂N-C-C-OH H₂C-C-H T CH3 Ο || Valine (Val) HO | || H₂N-C-C-OH !_ ______ € CH₂ CH₂ CH3 Methionine (Met) H 0 H₂N-C-C-OH H₂C H₂C-C-H CH3 Leucine (Leu) H 0 | || H₂N-C-C-OH CH₂ T SH Cysteine (Cys)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Valine ...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which amino acids in Table 17.1 have nonpolar R groups? Highly polar groups? Relatively flat R groups? Table 17.1 QUESTION CONTINUE TO NEXT PAGE Table 17.1 Names and Formulas of the Common Amino...
-
Which of the amino acids in Table 27.1 have more than one stereogenic center? Table 27.1 TABLE 27.1 -Amino Acids Found in Proteins (Continued) Abbreviation Structural formula* Name Amino acids with...
-
The debt of INKA consists of two bonds. The first bond was issued in 2011, with a 15-year maturity and a 6% coupon rate. The second bond was issued in 2016, with a 10-year maturity and a 8% coupon...
-
i. Find a. b. ii. Use the trapezium rule with 2 intervals to estimate the value of giving your answer correct to 2 decimal places. +6 e2x + 6 e2 dx,
-
In chronological order, the inventory, purchases, and sales of a single product for a recent month are as follows: Using the periodic inventory system, compute the cost of ending inventory , cost of...
-
For the current year ending March 31, Zing Company expects fixed costs of $425,750, a unit variable cost of $40, and a unit selling price of $65. a. Compute the anticipated break-even sales (units)....
-
In 1970, Rose Mary Knick purchased 90 acres of land in Scott Township, Lackawanna County, Pennsylvania. In 2008, another resident of Scott Township discovered documents that suggested that one of...
-
(Entries for Conversion, Amortization, and Interest of Bonds) Volker Inc. issued $2,500,000 of convertible 10-year bonds on July 1, 2010. The bonds provide for 12% interest payable semiannually on...
-
There are 15 exceptions to carrier liability under the Carriage of Goods by Sea Act other than "perils of the sea". Which ones are similar to an event of force majeure that are usually placed in a...
-
The amino acid alanine has the condensed structural formula shown here: Determine the VSEPR geometry about each internal atom and make a three-dimensional sketch of the molecule. NH,CH(CH3)COOH
-
What is the difference between a codon and a nucleotide? A codon and a gene?
-
On January 1, 2021, Power Ltd. issued bonds with a maturity value of $5 million for $4,797,000, when the market rate of interest was 8%. The bonds have a contractual interest rate of 7% and mature on...
-
In 2005, Doctora's Regional Hospital's Chief Executive Office (CEO) conducted performance reviews of the hospital's departments. The chief of oncology proposed increasing the number of patients (for...
-
The company is experiencing cash flow problems and is considering a change in credit terms which should increase sales and ultimately improve cash flows. In order to implement it successfully, they...
-
The glass in a window is 35 inches wide and 20 inches tall, and standard atmospheric pressure is 14.7 pounds per square inch. What net force would result in the glass if the air pressure outside the...
-
Providers are responsible for submitting accurate claims. Claims need to follow state and federal laws. Providers must follow guidelines to be reimbursed for the services that they provide. Providers...
-
Wall-e is stranded on a patch of frictionless surface (thanks to Mo the cleaning robot). Eva flies in with velocity vo, hits Wall-e and the two got entangled together. If the mass of Wall-e is Mw and...
-
What song best represents your experience in this class? State why by providing a description of the song or a few lyrics of the song.represents your experience in this class? State why by
-
What is an access control list?
-
Light from the star Vega has an intensity of about 2 x 10 8 W/m 2 . If Vega emits radiation with the same power as our Sun, how far is Vega from the Earth?
-
Unpolarized light with an electric field amplitude of 0.25 V/m is incident on a polarizer. What is the electric field amplitude of the transmitted light?
-
Linearly polarized light propagating along the y-direction is incident on a polarizer whose axis is parallel to the z-direction. If the intensity of the transmitted light is equal to 35% of the...
-
HYPOTHETICAL You work at a law firm, and you have been instructed by your supervising attorney to determine if the firm's client, Peterson, would be able to file and win a lawsuit against Electric...
-
FACT SCENARIO You are involved in a legal dispute over the home renovations done on the homeowners' property, which is a heritage home in which one of the parties grew up and holds great sentimental...
-
Sixty years later, is Tybee bomb still lurking? BY J.R. ROSEBERRY TYBEE BOMB TIMELINE " Feb. 5, 1958: An F-86 fighter jet collided with a B-47 bomber carrying a 7,600-pound Mark 15 nuclear bomb...

Study smarter with the SolutionInn App


