Which gas sample has the greatest pressure? Assume that all the samples are at the same temperature.
Question:
Which gas sample has the greatest pressure? Assume that all the samples are at the same temperature. Explain.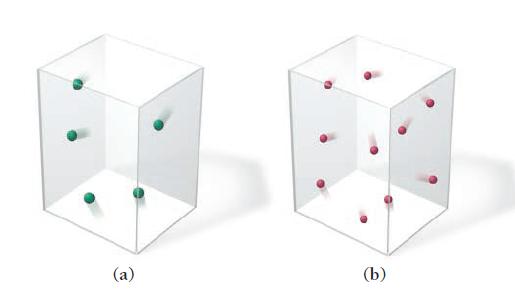
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider a 1.0-L sample of helium gas and a 1.0-L sample of argon gas, both at room temperature and atmospheric pressure. a. Do the atoms in the helium sample have the same average kinetic energy as...
-
Consider two gases, A and B, each in a 1.0- L container with both gases at the same temperature and pressure. The mass of gas A in the container is 0.34 g, and the mass of gas B in the container is...
-
Which gas sample has the greatest volume at STP? a) 10.0 g Ar b) 10.0 g Kr c) 10.0 g Xe d) None of the above (They all have the same volume.)
-
Consider a tank containing a liquid, and the rate of change of the liquid's height (h) with respect to time (t) is proportional to the difference between the current height and a reference height....
-
The Sorrento Hotel is a four-star hotel located in downtown Seattle. The hotels operations vice president would like to replace the hotels antiquated computer terminals at the registration desk with...
-
Replacement of a machine, income taxes, sensitivity. (CMA, adapted) The Smacker Company is a family-owned business that produces fruit jam. The company has a grinding machine that has been in use for...
-
Can you create a graphic of the Fairmont payroll systems company expenses that highlight general accounting clerk, Mary Perez, has company expenses for FICA (social security), Medicare and 401K,...
-
Donna Shader, manager of the Winter Park Hotel, is considering how to restructure the front desk to reach an optimum level of staff efficiency and guest service. At present, the hotel has five clerks...
-
You have just been named Director of Data Administration of General Hardware Co. General Hardware maintains a large central IS organization with several operational relational databases at its...
-
This picture represents a sample of gas at a pressure of 1 atm, a volume of 1 L, and a temperature of 25 C. Draw a similar picture showing what would happen to the sample if the volume were reduced...
-
A wine-dispensing system uses argon canisters to pressurize and preserve wine in the bottle. An argon canister for the system has a volume of 55.0 mL and contains 26.0 g of argon. Assuming ideal gas...
-
Gavin Products uses a perpetual inventory system. For 2018 and 2019, Gavin has the following data: Required: 1. For each year, compute cost of goods sold, the cost of ending inventory , and gross...
-
You are a student in a masters in health administration program and have recently started your internship in a large, urban health system. Your preceptor has asked you to write a job description for...
-
Robert Casey received his masters in health administration from a major healthcare management program ten years ago. Through a series of increasingly responsible positions in various healthcare...
-
For the past 20 years, Metropolitan Hospital celebrated the fact that 50 percent of its new hires in management positions had been women. The hospital assumed that with such a practice, women would...
-
Assume you are a manager at a low budget healthcare setting (e.g., local health department). What will you do to recruit new staff and to motivate current employees when competitors in the area are...
-
Hospital D has added a new process to its operations. The hospital has an excellent health and safety record, but its safety officer has just retired. Three weeks after the new process is...
-
The Byrd Company had the following transactions during 2010 and 2011: 1. On December 24, 2010, a computer was purchased on account from Computers International for $60,000. Terms of the sale were...
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
Using Castiglianos theorem and determine the horizontal deflection at C. EI is constant. There is a pin at A. Assume C is a roller and B is a fixed joint. 400 lb/ft 6 ft 10 ft 45
-
Use the method of virtual work and determine the horizontal deflection at C. EI is constant. There is a pin at A. Assume C is a roller and B is a fixed joint. 400 lb/ft 6 ft 10 ft 45
-
Using Castiglianos theorem and determine the horizontal deflection at C. The cross-sectional area of each member is indicated in the figure. Assume the members are pin connected at their end points....
-
recommend a price and marketing strategy for the newly established automobile manufacturer seeking to enter the market for specialist competition motorcycles.compare your recommendation to those for...
-
How can organizational leaders foster a culture that encourages and sustains advanced OCB among employees?
-
The state is beginning a new apprenticeship program for incarcerated individuals. Officials designed the program to improve the chances of inmates finding a job upon release and to increase the wages...

Study smarter with the SolutionInn App


