Which molecule exhibits optical isomerism? H (a) H-C-cl Br (b) Br H H | | C-C-H cl
Question:
Which molecule exhibits optical isomerism?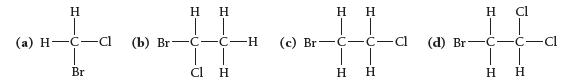
Transcribed Image Text:
H (a) H-C-cl Br (b) Br H H | | C-C-H cl H H H | | (c) Br C-c-c J-H CIH H Η (d) Br H 1 C-H ♫. ICIH C-C-Cl H Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
b This structure is the only one th...View the full answer

Answered By

Sandip Nandnawar
I am a B.E (Information technology) from GECA and also have an M.C.M from The University of RTMNU, MH.
I worked as a software developer (Programmer and TL). Also working as an expert for the last 6 years and deal with complex assessment and projects. I have a team and lead a team of experts and conducted primary and secondary research. I am a senior software engg and senior expert and deal with all types of CSE and IT and other IT-related assessments and projects and homework.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Obtain a Lewis formula of H2N2. This molecule exhibits isomerism (it has two isomers). Sketch the electron-dot formulas of these isomers. Describe the bonding in terms of valence bond theory, and...
-
Out of the following, the alkene that exhibits optical isomerism is (a) 2-methyl-2-pentene (b) 3-methyl-2-pentene (c) 4-methyl-1-pentene (d) 3-methyl-1-pentene
-
Draw the formula of an unsaturated bromide, C5H9Br, that can show a. Neither cis-trans isomerism nor optical activity b. Cis-trans isomerism but no optical activity c. No cis-trans isomerism but...
-
Use the Ratio Test to determine the values of x 0 for which each series converges. 00 X 2k 2 k=1 k
-
Tony Garcia opened a small dryer repair shop, Garcia Repair Shop, on January 2, 2010. The shop also sells a limited number of dryer parts. In January 2011, Garcia realized he had never filed any tax...
-
What is the difference between a bond and a note? How do the accounting treatments differ?
-
Compare and contrast the ethical approaches of its legal, therefore, its ok and the ends justify the means. Are there similarities? Are there differences?
-
Kogan Company has an inexperienced accountant. During the first 2 weeks on the job, the accountant made the following errors in journalizing transactions. All entries were posted as made. 1. A...
-
TheOnlyWizKid, Montclair State University Download and connect to the raw data Market Caps of S_P500 Companies 2008-2015.xls on Canvas>Files> Tableau Data Files using Tableau. The Excel file lists...
-
Name this alkane: CH-CH-CH-CH-CH-CH-CH-CH3 CH I CH3 CH3 CH3
-
Which property of carbon is related to its ability to form a large number of compounds? a) Its tendency to form four covalent bonds b) Its ability to form double and triple bonds c) Its tendency to...
-
Given n Z+, let the set M(n, k) Zn2 contain the maximum number of code words of length n, where the minimum distance between code words is 2k + 1. Prove that (The upper bound on |M(n, k)| is called...
-
Shan is a capital and income beneficiary of the Shan Family Trust which is an intervivos trust created by his father for him. The Shan Family Trust's income was $40,000 in gross capital gains in...
-
Selling price is $25 per unit and variable costs are $16 per unit. Calculate the contribution margin per unit. $1.56 $41.00 $11.00 $9.00King Ltd's contribution margin per unit is $2 and the...
-
How does software knowledge benefit your future endeavors as an accounting professional?
-
James Kolen, an employee of Smart Touch Learning, earned $128,600 prior to December. Kolen's salary for December is $10,000. Kolen authorized a $180 payment for health insurance and a $20...
-
A local retailer has Accounts Payable of $46,700 and Cost of Goods Sold of $355,000. Calculate their days' payable outstanding. Assess what the calculated days' payable outstanding implies about the...
-
Kingston, Inc., is looking to add a new machine at a cost of $4,432,455. The company expects this equipment will lead to cash flows of $801,456, $905,489, $919,442, $953,644, $1,207,987, and...
-
What are the principal differences among asset liquidity management, liability management, and balanced liquidity management?
-
An irregular conductor containing an irregular, empty cavity carries a net charge Q. (a) Show that the electric field inside the cavity must be zero. (b) If you put a point charge inside the cavity,...
-
You measure the electric field strength at points directly above the center of a square plate carrying charge spread uniformly over its surface. The data are tabulated in the next column, with x the...
-
A point charge -q is at the center of a spherical shell carrying charge +2q. That shell, in turn, is concentric with a larger shell carrying -3/2 q. Draw a cross section of this structure, and sketch...
-
To what extent does statute 1515(c) provide lawyers protection they otherwise would not have? Is this protection needed? Is it significant that statute 1515(c) was on the books for 14 years before...
-
As a department store manager, you are responsible for inventory management throughout the year. Explain how you would approach inventory management across the variation of the seasons. Your answer...
-
Anderson's backup argument was that the payments were gratuities rather than bribes. What is the significance of this argument? Why would anderson prefer that that payments be classified as...

Study smarter with the SolutionInn App


