Which sample of an ideal gas has the greatest pressure? Assume that the mass of each particle
Question:
Which sample of an ideal gas has the greatest pressure? Assume that the mass of each particle is proportional to its size and that all the gas samples are at the same temperature.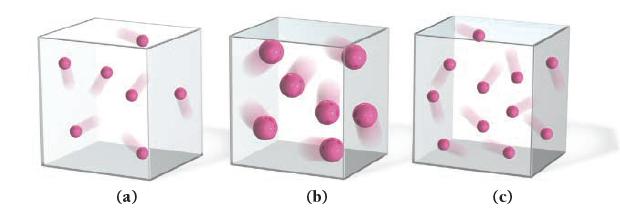
Transcribed Image Text:
(a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
c Since the temperature and the volume are both consta...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(Multiple choice) (1) The temperature change of two blocks of masses MA and MB is the same when they absorb equal amounts of heat. It follows that the specific heats are related by (a) cA =...
-
The town of Beautified is home to many corporations. In order to make sure that it establishes a fair competitive environment for all the City Council wants to pass a law Unfair methods of...
-
Which of the following is not true about an ideal gas? (a) The average kinetic energy of the gas molecules increases as the temperature increases. (b) The volume of an ideal gas increases with...
-
Prepare an Orientation Plan for the select position utilizing the theories discussed in the text. Discuss appropriate and effective orientation plans and your decision-making process for selecting a...
-
Continuous auditing has the potential to reduce labor costs associated with auditing. It also can provide audit assurance closer to the occurrence of a transaction, which improves the reliability of...
-
Two bonds have the following features: The structure of yields is Term Interest Rate 1 year .....6% 2 years .....7% 3 years .....8% 4 years .....9% 5 years .....10% a) What is the valuation of each...
-
A & R Industrial Supply shows the following financial statement data for 2008, 2009, and 2010. Prior to issuing the 2010 statements, auditors found that the ending inventory for 2008 was understated...
-
In a production facility, 1.2-in-thick, 2-ft = 2-ft square brass plates (r = 532.5 lbm/ft3 and cp = 0.091 Btu/lbm ¢ °F) that are initially at a uniform temperature of 75°F are heated by...
-
Provide a brief review of the following research paper: https://www.sciencedirect.com/science/article/abs/pii/S1566014120301072
-
A sample of gas has a mass of 0.311 g. Its volume is 0.225 L at a temperature of 55 C and a pressure of 886 mmHg. Find its molar mass. SORT The problem gives you the temperature and pressure of...
-
Explain why people may experience ear pain after a rapid change in altitude.
-
In Problems 23 34, graph each system of linear inequalities. 2x + y 2 -2 2x + y 2 2
-
How would ask for funds from an investor? What are the difference between pre-money valuation and post-money valuation? Illustrate the final answer on an expandable pie. Number of shares = 2,000,000...
-
Identify the history of assessment in counseling. Describe the importance of assessment in counseling and the role of assessment in the role of school counseling.
-
The derivative of an unknown function f(x) is shown below. f'(x) = (2x-1)(x-3) Note that the following questions are about the original function f(z). (a) Determine all critical points of f(x), and...
-
On January 1, Chadd has located a few private investors that wish to purchase shares in the new corporation. Some want common shares, while other are interested in preferred shares. Gescan Electric...
-
In today's business world, do you see any potential downside to using a strong power-oriented linguistic style when leading others? If no, why? If yes, what are they? I belive that there is no down...
-
Following are the balance sheets at year-end 2009 and 2010 followed by the 2010 income statement of Sactor Inc. The annual report for year 2010 provides the following supplemental information: 1. The...
-
Representative data read from a plot that appeared in the paper Effect of Cattle Treading on Erosion from Hill Pasture: Modeling Concepts and Analysis of Rainfall Simulator Data (Australian Journal...
-
Apply the moment-distribution method to determine the moment at each joint of the symmetric parabolic haunched frame. Supports A and D are fixed. Use Table 132. The members are each 1 ft thick. E is...
-
Determine the horizontal and vertical displacements at joint ¢of the assembly in Prob. 141. Data From Prob. 141. Determine the stiffness matrix K for the assembly. Take A = 0.5 in 2 and E =...
-
Determine the horizontal displacement of joint and the force in member ¡ Take A = 0.75 in 2 , E = 29(10 3 ) ksi. 2 500 lb 4 ft (3) -3 -31H 4 ft-
-
You are analyzing an airplane with a weight of 11,000 lb, wing area of 280 ft, wing span of 46 ft, and a CD,o of 0.027. The airplane is cruising at an altitude of 20,000 ft a. Construct a graph of...
-
A new pipeline system is designed to carry non-sour natural gas from a compressor station 1 to three distribution tie-ins in a power plant fuel station. The pipeline route is given in the following...
-
5. Block stacking is used for the storage of 48" x 48" unit loads. A counterbalance lift truck is used to store and retrieve the unit loads, which are stacked five high. The storage aisle is 13'...

Study smarter with the SolutionInn App


