Which set of four quantum numbers corresponds to an electron in a 4p orbital? a) n =
Question:
Which set of four quantum numbers corresponds to an electron in a 4p orbital?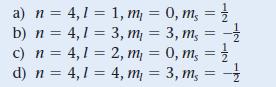
Transcribed Image Text:
a) n = 4,1 = 1, m₁ = 0, m, = // b) n = 4,1 = 3, m₁ = 3, m, = -1/ c) n = 4,1 = 2, m₁ = 0, m, = // d) n = 4,1 4, m₁ = 3, m = - =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a n 4...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which set of four quantum numbers corresponds to an electron in a 4d orbital? n = 4, l = 2, m = 1, m, n = 4, l = 1, m = 0, ms n = 4,1 = 0, m = 3, ms n = 4,1 = 3, m = 0, ms Submit Request Answer ||...
-
1. (1pt) Use Cisco Packet Tracer to create the network shown above. Submit a screenshot of the network your created. 2. (1pt) Configure the hostnames, IP addresses, subnet masks, and default gateways...
-
Using an orbital box diagram and noble gas notation, show the electron configuration of titanium. Give one possible set of four quantum numbers for each of the electrons beyond those of the preceding...
-
List and describe the payment options (terms of sale) that can be applied to domestic U.S. shipments.
-
Peavler Corporation issues $400,000 of 9%, 5-year bonds on January 1, 2010, at 104. If Peavler uses the effective-interest method in amortizing the premium, will the annual interest expense increase...
-
What is the focus of the standards of field work for attestation and audit engagements? a. Guidelines for training, proficiency, and due care. b. Guidelines for the content of the practitioners...
-
In quantum mechanics, what does it mean for two things to be in a state of entanglement?
-
The CAB Partnership, although operating profitably, has had a cash flow problem. Unable to meet its current commitments, the firm borrowed $34,000 from a bank giving a long-term note. During a recent...
-
7. Scotty Quadcopters plans to sell a standard quadcopter (toy drone) for $65 and a deluxe quadcopter for $95. Scotty purchases the standard quadcopter for $45 and the deluxe quadcopter for $70....
-
Julio and Milania are owners of Falcons Corporation, an S corporation. They each own 50 percent of Falcons Corporation. In year 1, Julio and Milani received distributions of $20,000 and $10,000,...
-
A main-group element has an outer electron configuration of ns 2 np 4 . What charge is likely for an ion of this element? (a) 1- (b) 2- (c) 1+ (d) 2+
-
Who is credited with arranging the periodic table? How are the elements arranged in the modern periodic table?
-
Write an application that displays a series of at least eight student ID numbers (that you have stored in an array) and asks the user to enter a test letter grade for the student. Create an Exception...
-
Current Attempt in Progress You are presented with the following alphabetical list of accounts and balances (in thousands) for Crane Enterprises Inc. at June 30, 2024. All accounts have a normal...
-
Cash $3,240 Accounts Receivable 3,924 Supplies 3,672 Prepaid Insurance 1,800 Equipment 7,680 Accum. Depr. - Equipment $1,296 Accounts Payable 1,014 Long Term Notes Payable 8,400 Common Stock 2,400...
-
13-40. Cycle-1 is a fast-growing start-up firm that manufactures bicycles. The following income statement is available for October: Sales revenue (300 units @ $600 per unit) Less Manufacturing costs...
-
List five items that should appear on a customer's Accounts Receivable Ledger.
-
The general purpose of producing a trial balance is to ensure that the entries in a company's bookkeeping system are mathematically correct. A trial balance is so called because it provides a test of...
-
Explain the major characteristics of an equity-indexed annuity.
-
For the following arrangements, discuss whether they are 'in substance' lease transactions, and thus fall under the ambit of IAS 17.
-
What is the solution of the time-dependent Schrdinger equation (x,t) for the total energy eigenfunction (x) = 2/a sin (3x/a) for an electron in a one-dimensional box of length 1.00 10 10 m? Write...
-
Derive an equation for the probability that a particle characterized by the quantum number n is in the first 25% (0 x a/4) of an infinite depth box. Show that this probability approaches the...
-
It is useful to consider the result for the energy eigenvalues for the one-dimensional box E n = h 2 n 2 /8ma 2 n = 1, 2, 3 , . . . as a function of n, m, and a. a. By what factor do you need to...
-
Assume that you are the president of Highlight Construction Company. At the end of the first year of operations (December 31), the following financial data for the company are available: Cash...
-
Compose a 400-500 word message to your audience of college students briefly teaching them about plagiarism, its consequences, and its impact on a writer's authority. Your educational message should...
-
Explain a real-life incident that happened to you or an experience you had that prompts you to write an argumentative response. Think of a time when you had a problem, issue, or argument at school,...

Study smarter with the SolutionInn App


