Would you expect an alloy of iron and vanadium to be substitutional or interstitial? Explain your choice.
Question:
Would you expect an alloy of iron and vanadium to be substitutional or interstitial? Explain your choice. Create a phase diagram for the binary alloy of iron and vanadium based on the tabulated data provided.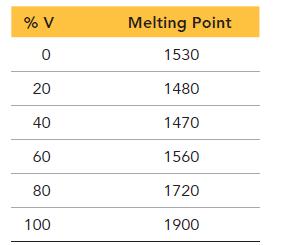
Transcribed Image Text:
% V 0 20 40 60 80 100 Melting Point 1530 1480 1470 1560 1720 1900
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Based on the melting point data provided in the image I would expect an alloy of iron and vanadium t...View the full answer

Answered By

HABIBULLAH HABIBULLAH
I have been tutor on chegg for approx 5 months and had solved a lot of questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An alloy of iron and carbon was treated with sulfuric acid, in which only iron reacts. 2Fe(s) + 3H2SO4(aq) Fe2(SO4)3(aq) + 3H2(g) If a sample of alloy weighing 2.358 g gave 0.1152 g of hydrogen,...
-
An alloy of iron (54.7%), nickel (45.0%), and manganese (0.3%) has a density of 8.17 g/cm3. How many iron atoms are there in a block of alloy measuring 10.0 cm 20.0 cm 15.0 cm?
-
The binary phase diagram for the silver (Ag) and germanium (Ge) system is shown in Figure 11-34. Figure 11-34 The silver-germanium phase diagram (for Problem 11-35). (a) Schematically draw the phase...
-
You find a certain stock that had returns of 18 percent, 23 percent, 16 percent, and 9 percent for four of the last five years. If the average return of the stock over this period was 10.3 percent,...
-
Johson Corporation issued bonds twice during 2010. The transactions were as follows: 2010 Jan 1Issued $1,000,000 of 7.5 percent, 10-year bonds dated January 1, 2010, with interest payable on June 30...
-
From the following list, identify the accounts that should be closed to Income Summary at the end of the fiscal year under a perpetual inventory system: (a) Accounts Payable, (b) Advertising Expense,...
-
Monroe Bradstad borrowed \($100,000\) from his aunt, Jeanne Garland, to purchase farmland. Both parties subsequently signed a promissory note stipulating that interest would be accrued prior to or on...
-
The following items were selected from among the transactions completed by Aston Martin Inc. during the current year: Apr. 15. Borrowed $225,000 from Audi Company, issuing a 30-day, 6% note for that...
-
The given code starts by initializing some sets and functions f , g , and h . Block 1 is a function ( called hasInverse ) that takes as input another function ( called func ) , and two sets ( D and T...
-
Create a table comparing and contrasting pyrometallurgy, hydrometallurgy, and electrometallurgy. Include a definition, an application or two, and any relevant notes for each type of metallurgy. Once...
-
Assign each element in the first row of the transition metals, scandium through zinc, to a group member. Have each group member look up on the Internet or in a reputable chemistry reference book the...
-
What are the dimensions of each of the following matrices? a. [7] b. c. [4 10 17 15 11] d. e. -3 3
-
If f(x) = 4x(sin x + cos x), find f'(x)= f'(5) =
-
ECs regular tax depreciation for the year is correctly calculated as $350,000 before considering the current year fixed asset additions of $840,000 (see table below). EC wants to claim the fastest...
-
What is the central theme and subject matter addressed in Naomi Wolf's literary work "Misconceptions"?
-
The Sieve of Eratosthenes, invented by the Greek mathematician of the same name, provides a quick way to nd all prime numbers within a given range. The algorithm involves creating an array of bytes...
-
A firm pays a current dividend of $1, which is expected to grow at a rate of 9% indefinitely. If the current value of the firm's shares is $109, what is the required return applicable to the...
-
For each of the following scenarios, indicate the Bad Debt Expense to be recorded in 2011, the balance in the Allowance for Doubtful Accounts account at December 31, 2011, and the net realizable...
-
Identify the Critical Infrastructure Physical Protection System Plan.
-
Two particles with the same charge are attached to strings that are 80 cm long and that hang from a ceiling as shown in Figure P17.97. If the angle between the strings is ? = 60? and the particles...
-
Consider three particles of charge Q, 2Q, and 4Q, where Q = 2.4 C, arranged as shown in Figure P17.98. If a particle of charge q = -1.5 C is placed at the origin,? (a) What are the components of the...
-
If the electric field is zero in a particular region of space, what does that tell you about the electric potential in that region? Is the potential zero, constant, or something else? Explain.
-
beta of Stock R 1.1 nominal risk-free rate 4% market risk premium 7% Under CAPM, the required return on Stock R is:
-
2. An electron neutrino is produced in a nuclear reaction in the sun, with an energy E = 200 KeV. (a) In terms of the neutrino mass eigenstates V1), 2), describe the wave function of the neutrino...
-
Consider a polytropic model for a star, in which so a star with Yp is an "n = = Yp P(r) = Kp(r) = Kp(r)1+1/n = = 4/3 5/3 is an "n 3/2 polytrope", and one with Yp 3 polytrope. (Here n is an index, not...

Study smarter with the SolutionInn App


