Question: Write Lewis structures for each molecule or ion. Use expanded octets as necessary. a. C. CIFS Cl3PO b. AsF6 d. IFs
Write Lewis structures for each molecule or ion. Use expanded octets as necessary.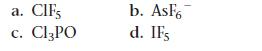
a. C. CIFS Cl3PO b. AsF6 d. IFs
Step by Step Solution
★★★★★
3.50 Rating (153 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

a F F Chlorine pe... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


