An element X has five major isotopes, which are listed below along with their abundances. What is
Question:
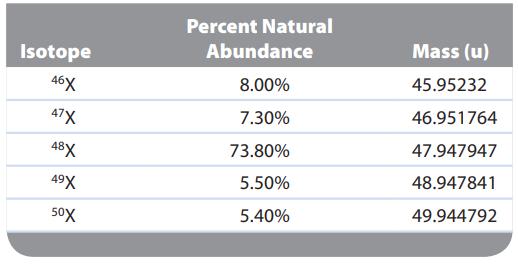
An element “X” has five major isotopes, which are listed below along with their abundances. What is the element?
Transcribed Image Text:
Isotope 46X 47X 48X 49X 50X Percent Natural Abundance 8.00% 7.30% 73.80% 5.50% 5.40% Mass (u) 45.95232 46.951764 47.947947 48.947841 49.944792
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Isotopes Those elements which have the same atomic ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
An element X has five major isotopes, which are listed below along with their abundances. Calculate the average atomic mass, and identify the element. Isotope Percent Natural Abundance 8.00 7.30...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Roll Board Inc. manufactures several models of high-quality skateboards. The companys ABC system has four activity cost pools, which are listed below along with their activity measures and activity...
-
Let Z[] be the following set of rational numbers { a Z, k N} (and recall that N = {0, 1, 2,...} in this class). Let the addition and multiplication for Z[] be the usual addition and multiplication...
-
Health Drinks Company produces two beverages, PowerPunch and SlimLife. Data about these products follow. Additional data from its two production departments follow. Required 1. Determine the cost of...
-
Briefly define the product life cycle and identify the life phases the product experiences.
-
LO7 How does the wherewithal-to-pay concept affect the tax treatment of prepaid income?
-
The Pilot Pen Company has decided to use 15 test markets to examine the sensitivity of demand for its new product to various prices, as shown in the following table. Advertising effort was identical...
-
Overhead Variance (Over or Underapplied), Closing to cost of Goods Sold At the end of the year, Estes Company provided the following actual information Overhead 5412,600 Direct Labor cost 532,000...
-
5,000 cubic feet per minute (CFM) of 25F outside air mixes with 70F return air. The mixed air is heated to 95F as it passes across the heating coil, then across the cooling coil, before being...
-
What mass of silver chloride can be prepared by the reaction of 100.0 mL of 0.20 M silver nitrate with 100.0 mL of 0.15 M calcium chloride? Calculate the concentrations of each ion remaining in...
-
Which of the following pairs of compounds have the same empirical formula? a. acetylene, CH, and benzene, C6H b. ethane, CH6, and butane, C4H10 c. nitrogen dioxide, NO2, and dinitrogen tetroxide, NO4...
-
What is the present value of the following uneven cash flow stream? The annual interest rate is10%. 4 Years 300 300 100 -50 4. 3. 2.
-
Discuss how Central Governments Exercise Control over Local Governments by Citing Examples
-
Find derivative of sin x,cos a,tan x,secx,csca, cot a
-
Find anti derivative of cos x,sin x,sec r,sec r tan r,csc xcotx,csc r
-
Find derivative of arcsin x
-
1. Examine the impact of feedwater heater pressure on cycle efficiency and determine the intermediate pressure for optimal performance for one heater.
-
What is meant by the designation M20 2.5-6g6g? (What does the "" mean?)
-
For liquid water the isothermal compressibility is given by; where r and b are functions of temperature only. If 1 kg of water is compressed isothermally and reversibly from I to 500 bar at 60(C. how...
-
Which of the following pure compounds will exhibit hydrogen bonding? a) CH 3 CH 2 OH b) CH 2 O c) C 2 H 4 d) C 2 H 2 e) CH 3 OCH 3 f) CH 3 NH 2 g) C 3 H 8 h) NH 3
-
For each case below, identify the most likely value for x: a) BH x b) CH x c) NH x d) CH 2 Cl x
-
Identify the hybridization state and geometry of each carbon atom in the following compounds: a. b. c. -OEJ- -CEC-C . .C. H. 1 H' .
-
Ron (78) is single and retired many years ago. during the tax year, he received a federally taxable private pension distribution of $19563. His Ohio income tax base is $55194. What is the amount of...
-
On January 1, 2020, Victory Co. acquired 60% of the shares of Sauce Ltd. by issuing shares valued at $1,500,000. On this date, Sauce Ltd.'s building and machinery had remaining useful lives of 10...
-
Anderson Co. issued a $59,335, 60-day, discounted note to National Bank. The discount rate is 7%. At maturity, assuming a 360-day year, the borrower will pay a.$60,027 b.$59,335 c.$55,182 d.$58,643

Study smarter with the SolutionInn App


