Arrange the atoms and/or ions in the following groups in order of decreasing size. a. 0, 0,
Question:
Arrange the atoms and/or ions in the following groups in order of decreasing size.
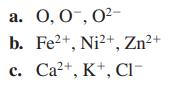
Transcribed Image Text:
a. 0, 0, 0²- b. Fe²+, Ni²+, Zn²+ c. Ca²+, K+, Cl-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a In this group all the species are oxygen ions To arrange ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
2019A Company 1. The following information is taken from the records of 2019A Company for July: Purchases Direct materials Sales Salaries and wages: Selling and administrative salaries Direct...
-
40. Select the most acidic compound from the following. A. SO 2 B. Al 2 O 3 C. CaO D. PbO E. H 2 O 41. Select the most basic compound from the following. A. Bi 2 O 3 B. SiO 2 C. Cs 2 O D. Na 2 O E. H...
-
Arrange the following atoms in order of decreasing atomic radius: Na, Al, P, Cl, Mg.
-
You name the catastrophe, and JIT has been through it and survived. Toyota Motor Corporation has had its world-renowned JIT system tested by fire. The massive fire incinerated the main source of...
-
Is a current account deficit always associated with a strong real exchange rate (that is, one in which the currency is overvalued compared to the PPP prediction)?
-
(a) Briefly describe the phenomenon of coring and why it occurs. (b) Cite one undesirable consequence of coring.
-
MAC Industries completed the following inventory transactions during the month of August: Requirements 1. Without resorting to calculations, determine which inventory method will result in MAC...
-
A firm pays a $1.50 dividend at the end of year one (D1), has a stock price of $155 (P0), and a constant growth rate (g) of 10 percent. a. Compute the required rate of return (Ke). Indicate whether...
-
Ankh - Sto Associates Co . is expected to generate a free cash flow ( FCF ) of $ 4 , 8 3 5 . 0 0 million this year ( FCF = $ 4 , 8 3 5 . 0 0 million ) , and the FCF is expected to grow at a rate of 2...
-
Which of the following would you expect to be more favorable energetically? Explain. a. An H 2 molecule in which enough energy is added to excite one electron from the bonding to the antibonding MO...
-
Classify the bonding in each of the following molecules as ionic, polar covalent, or nonpolar covalent. a. H 2 b. K 3 P c. NaI d. SO 2 e. HF f. CCl 4 g. CF 4 h. K 2 S
-
Use Theorem 3.11 to evaluate the following limits. THEOREM 3.11 Trigonometric Limits sin x lim X-0 X 1 lim x-0 COS X x = 0
-
Why do you think a debt instrument whose interest rate is changed periodically based on some market interest rate would be more suitable for a depository institution than a long-term debt instrument...
-
Explain the objectives and functions of the World Trade Organization.
-
Devise a business plan to promote your organization overseas: a. What are the unique attributes that your organization can bring to an overseas market? b. How would you conduct a needs analysis of...
-
What is meant by: a. individual banking b. institutional banking c. global banking
-
Explain trade creation and trade diversion.
-
Do Men Have a Higher Mean Body Temperature? Repeat Exercise 9 with the additional assumption that 1 = 2. How are the results affected by this additional assumption? Assume that the two samples are...
-
A researcher reports a significant two-way between-subjects ANOVA, F(3, 40) = 2.96. State the decision to retain or reject the null hypothesis for this test.
-
Calculate the pH of a 0.010-M solution of iodic acid (HIO 3 , K a = 0.17).
-
A solution is tested for pH and conductivity as pictured below: The solution contains one of the following substances: HCl, NaOH, NH 4 Cl, HCN, NH 3 , HF, or NaCN. If the solute concentration is...
-
When determining the pH of a weak acid solution, sometimes the 5% rule can be applied to simplify the math. At what K a values will a 1.0-M solution of a weak acid follow the 5% rule?
-
Q: 9 Check whether (A + B)C = AC + BC where A= Ja and C = [15]. [10] Q: 10 Prove that for any square matrix A with real number entries, A + A' is a symmetric matrix and A - A' is a skew symmetric...
-
Q: 7 Letf: NY be a function defined as f (x) = 4x + 3, where, Y = {y N: y = 4x + 3 for somex N}. Show that f is invertible. Find the inverse. [10] Q: 8 Let f: X Y be a function. Define a relation R...
-
Q: 6 Find all the points of local maxima and local minima of the function f given by f(x)=2x-6x+6x+5.

Study smarter with the SolutionInn App


