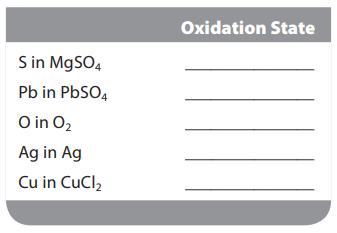
Assign the oxidation state for the element listed in each of the following compounds: S in MgSO4
Question:
Assign the oxidation state for the element listed in each of the following compounds:
Transcribed Image Text:
S in MgSO4 Pb in PbSO4 O in O₂ Ag in Ag Cu in CuCl₂ Oxidation State
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
1 S in MgSO4 Magnesium sulfate Mg is in Group 2 and typically has an oxidation state of 2 ...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
n i=1 - - let denote the sample correlation coefficient between Y and X (defined formally below), and let = 1 (yi ) and = - 1(xi). (These notations are also defined in slides 44-46 of Chapter 1...
-
Indicate the oxidation state for the element noted in each ssof the following: a. H in CaH2 b. H in H2O c. C in CH4 d. S in H2SO4
-
Indicate the oxidation state for the element noted in each of the following: a. H in H2 b. H in C2H4 c. Si in SiH4 d. N in HNO3
-
In Boolean algebra, the bar sign (-) indicates: Select one: a. NOT operation b. AND operation C. OR operation d. All of the mentioned
-
In conducting interviews and observing factory operations to implement an activity-based costing system, you determine that several activities are unnecessary or redundant. For example, warehouse...
-
Five years ago, Frater Zahns Company invested 38 million30 million in fixed capital and another d8 million in working capitalin a bakery. Today, Frater Zahns is selling the fixed assets for 21...
-
Identify the research scenario, including the general area of focus. Develop a hypothetical research scenario that would necessitate the use of the Action Research Approach and a Posttest With a...
-
The August 31 bank statement of Watson Healthcare has just arrived from United Bank. To prepare the bank reconciliation, you gather the following data: a. The August 31 bank balance is $5,630. b. The...
-
At the conclusion of a second-year practical session, two groups of students engage in a heated debate. Group A students are certain that their results (Table 1) are statistically comparable to those...
-
Balance sheets for P Company and S Company on August 1, 2019, are as follows: Required: Prepare a workpaper for a consolidated balance sheet for P Company and its subsidiary on August 1, 2019, taking...
-
Consider the reaction between oxygen (O 2 ) gas and magnesium metal to form magnesium oxide. Using oxidation states, how many electrons would each oxygen atom gain, and how many electrons would each...
-
Consider the reaction between sodium metal and fluorine (F 2 ) gas to form sodium fluoride. Using oxidation states, how many electrons would each sodium atom lose, and how many electrons would each...
-
Figure 43-15 shows an early proposal for a hydrogen bomb. The fusion fuel is deuterium, 2H.The high temperature and particle density needed for fusion are provided by an atomic bomb "trig- ger" that...
-
6 m Two point charges, +8 nanoC and +2 nanoC are separated by 6 meters. What is the magnitude of the electric field (in N/C) midway between them? +8 nC P4 3 m +2 nC
-
Heather is planning to invest a constant amount of money at the end of every year for 15 years and then allow her money to accumulate interest for 15 more years without any additional deposits. If...
-
The prospect of tourism sector is not so good during this COVID-19 pandemic period. Tasha believe that the share price of tourism related companies will fall as a result of lower earnings. One of the...
-
Consider the adaptive modulation system. There is a three-state channel: the SNR of the channel is 3dB for 20% of the time, 10dB for 40% of the time, and 17dB for 40% of the time. With the adaptive...
-
You have just inherited $200,000 and wisely decided to invest your money in a diversified portfolio with your investment firm. Your financial advisor tells you that you can earn an average of 8%...
-
Using data from the stockholders equity section of Tramot Corporations balance sheet that follows, and assuming one years dividend in arrears, compute the book value per share for both the preferred...
-
Propose a reasonable mechanism for the following reaction. OH
-
You isolate a compound with the formula PtCl 4 . 2KCl. From electrical conductance tests of an aqueous solution of the compound, you find that three ions per formula unit are present, and you also...
-
Which of following statement(s) is(are) true? a. Phosphoric acid is a stronger acid than nitric acid. b. The noble gas with the lowest boiling point is helium. c. Sulfur is found as the free element...
-
The atmosphere contains 9.0 10 -6 % Xe by volume at 1.0 atm and 25 C. a. Calculate the mass of Xe in a room 7.26 m by 8.80 m by 5.67 m. b. A typical person takes in about 2 L of air during a...
-
Brandon Technology makes two models of a specialized sensor for the aerospace industry. The difference in the two models relates to the required accuracy of the sensor. The Standard model is used for...
-
Functions f and h are invertible functions. f(x)=11-(x)/(4) and h(x)=-4(x-11) Answer two questions about these functions. Write a simplified expression for f(h(x)) in terms of x.
-
The cost for an eight-grade party is $450 for room rental , entertainment, and decorations, plus $20 per person for food. Tickets for the party are sold $25. What is the break-even point?

Study smarter with the SolutionInn App


