Consider the following electrostatic potential diagrams for some covalent compounds. Which of the represented compounds would not
Question:
Consider the following electrostatic potential diagrams for some covalent compounds. Which of the represented compounds would not be soluble in water?
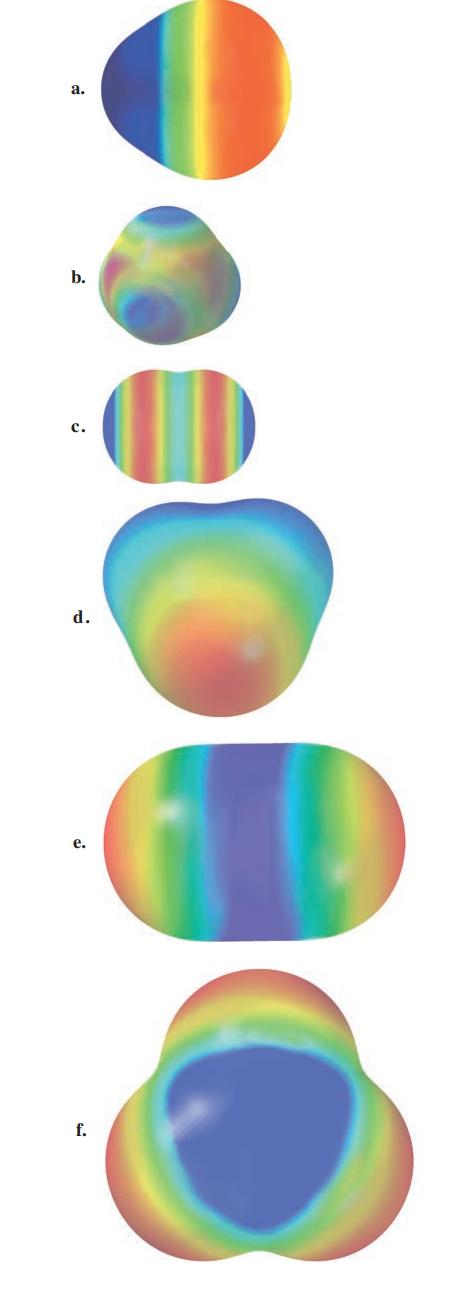
Transcribed Image Text:
a. b. C. d. e. f.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The compounds that are not soluble in water are a b c d e f and g Water is a polar molecule and it i...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Consider the following electrostatic potential diagrams: Rank the compounds from lowest to highest boiling point, and explain your answer. Ethanol Propane Acetone
-
The following electrostatic potential diagrams represent H2, HCl, or NaCl. Label each, and explain your choices. (a) (b)
-
The following electrostatic potential diagrams represent CH4, NH3, or H2O. Label each, and explain your choices. a. C. b.
-
You start driving east for 14 miles, turn left, and drive north for another 10 miles. At the end of driving, what is your straight line distance from your starting point? Round to the nearest tenth...
-
Kelly Malone plans to have $50 withheld from her monthly paycheck and deposited in a savings account that earns 12% annually, compounded monthly. If Malone continues with her plan for two and...
-
Hermann Corporation is considering an investment of 375 million with expected after-tax cash inflows of 115 million per year for seven years and an additional after-tax salvage value of h50 million...
-
Use information from Section 6.7 to estimate which form of electromagnetic radiation is the lowest energy ionizing radiation. Data from section 6.7 When we first introduced the concept of the...
-
On July 1, 2014, Sutton Inc. invested $720,000 in a mine estimated to have 800,000 tons of ore of uniform grade. During the last 6 months of 2014, 120,000 tons of ore were mined and sold....
-
1. Explain why all of the following values are equal. What is the value shown in each line below? 9-2 9-x-y 1 dzdydx 9-x2-y2 /9-y2 9-y2-22 2 1 dxdzdy 9-y2 8 3 9-x c9-xz 70 1 dydzdx
-
Tea has been in the limelight recently thanks to increasing global demand, especially in emerging economies like China and India. China, India, Kenya, and Sri Lanka are some of the largest producers...
-
Consider the steps involved in balancing oxidation reduction reactions by using oxidation states. The key to the oxidation states method is to equalize the electrons lost by the species oxidized with...
-
What is an acidbase reaction? Strong bases are soluble ionic compounds that contain the hydroxide ion. List the strong bases. When a strong base reacts with an acid, what is always produced? Explain...
-
Find the interpolating polynomial p(t) = (0 + (1t + a2t2 for the data (1,6), (2,15), (3, 28). That is, find (0, (1; and (2 such that 2-6 + ai (l)+a2 ( 1 ) ap a1 (2) +a2(2)15 + ai (3) + a2(3)2-28
-
Initial Conditions In physics, there are special states that we care about. One of these is the initial conditions of a problem. This is just the answer to the question "what are the position and...
-
Assume that the Oakley Corporation has fixed costs of $100,000 and variable costs of $100 per unit. Which selling prices will yield the maximum net income?
-
Katherine owns several apartment buildings in corvallis, oregon, that she rents out to university students. she uses part of the rental income to pay for insurance premiums. how should katherine...
-
Baker v. Carr (1962) saw the Supreme Court enter a "political thicket" that it had been careful to avoid in the past. However, the politics of the decision extend beyond the fact that it dealt with...
-
Eloise Corp. produces an unfinished toy, which currently sells for $60. To produce this unfinished toy, it costs Eloise Corp. $20. Recently, Eloise thought about continuing processing the toy by...
-
Mazurka Corporation has 2,000 shares of $100, 8 percent cumulative preferred stock outstanding and 40,000 shares of $1 par value common stock outstanding. In the companys first three years of...
-
A 20-cm-square vertical plate is heated to a temperature of 30oC and submerged in glycerin at 10oC. Calculate the heat lost from both sides of the plate.
-
Consider element 113, Nh. What is the expected electron configuration for Nh? What oxidation states would be exhibited by Nh in its compounds?
-
Many lithium salts are hygroscopic (absorb water), but the corresponding salts of the other alkali metals are not. Why are lithium salts different from the others?
-
There are three known xenon fluoride covalent compounds: XeF 2 , XeF 4 , and XeF 6 . In general, the xenon fluoride compounds must be stored in an inert atmosphere, free of oxygen and water. Why is...
-
Janice mixes her own bird seed. She recently purchased 7/8pounds of sunflower seed, 7-1/2 pounds of cracked corn, and 6-5/8pounds of millet. After mixing the seed, how many pounds of birdseed did she...
-
3. Your uncle runs a stand selling shaved ice cones near the Home Depot. He sells five flavors of shaved ice: cherry, grape, lime, orange and peppermint. He is considering discontinuing the least...
-
What is 20% of 90? Write your answer rounded to the nearest whole number. r30% of 240 is what number? Round your answer to the nearest whole number. r230% of what number is 460? Round your answer to...

Study smarter with the SolutionInn App


