For the preceding question, which of the following equations best represents the reaction? 4NH3 + 4N a.
Question:
For the preceding question, which of the following equations best represents the reaction?
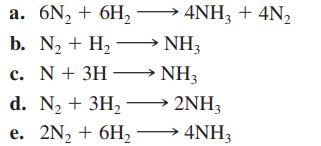
Transcribed Image Text:
4NH3 + 4N₂ a. 6N₂ + 6H₂ b. N₂ + H₂NH3 c. N + 3H NH3 d. N₂ + 3H₂ →→→ 2NH3 e. 2N₂ + 6H₂ 4NH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
The equation that best represents the reaction in the image is NHNOs HOl NHaq NOaq This equation sh...View the full answer

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Which of the following equations best represents the reaction shown in the diagram? (a) 8A + 4B C + D (b) 4A + 8B 4C + 4D (c) 2A + B C + D (d) 4A + 2B 4C + 4D (e) 2A + 4B C + D A B D
-
Which of the following equations best represents the reaction shown in the diagram? (a) A + B C + D (b) 6A + 4B C + D (c) A + 2B 2C + D (d) 3A + 2B 2C + D (e) 3A + 2B 4C + 2D ABCD
-
For the preceding question, which of the following equations best represents the reaction? a. 6N 2 + 6H 2 4NH 3 + 4N 2 b. N 2 + H 2 NH 3 c. N+ 3H NH 3 d. N 2 + 3H 2 2NH 3 e. 2N 2 + 6H 2 4NH 3...
-
Which of the following statements are true about REST? Pick ONE OR MORE options Logical URLs should be used instead of physical URLS Adal URLs must always be used in REST response A paging technique...
-
Crafton Manufacturing produces machine tools for the construction industry. The following details about overhead costs were taken from its company records. Additional information on the drivers for...
-
Define and explain the use of contribution margin and total contribution margin.
-
Although the customer loyalty project at Petrie Electronics had gone slowly at first, the past few weeks had been fast-paced and busy, Jim Watanabe, the project manager, thought to himself. He had...
-
(Conversion of Bonds) On January 1, 2009, Trillini Corporation issued $3,000,000 of 10-year, 8% convertible debentures at 102. Interest is to be paid semiannually on June 30 and December 31. Each...
-
A firm just paid an annual dividend of $3.50 per share. Dividends are expected to grow at 5% per year. If the discount rate for this firm is 12%, how much is a share of this firm worth?Group of...
-
It is important to ensure passenger comfort on ships by stabilizing the ship's oscillations due to waves [13]. Most ship stabilization systems use fins or hydrofoils projecting into the water to...
-
What is the difference between the empirical and molecular formulas of a compound? Can they ever be the same? Explain.
-
Nitrogen gas (N 2 ) and hydrogen gas (H 2 ) react to form ammonia gas (NH 3 ). Assuming the reaction goes to completion, draw a representation of the product mixture. Explain how you arrived at this...
-
What is the marketing concept? How can pricing activities be guided by the marketing concept?
-
The two-sided transfer function of a linear time-invariant (LTI) system, H(z), is described as H(z) = (1-2z-)(1+2) (1-0.2z-)(1-0.4z-) (a) Classify whether H(z) is maximum, minimum or mixed phase...
-
What is the output of the following code fragment? int x = 0; while( x < 8) cout < < x < < X ++; cout < < x < < endl; 11
-
Harrison and Gloria have been negotiating over the sale of bulk raw materials (lumber, iron and steel). Each party has had an army of lawyers acting on both sides trying to get the deal done and it...
-
35y2(x-3) (x+3) Simplify 21y (x-3)
-
What is one of the primary objectives of the PCI DSS (Payment Card Industry Data Security Standard)?
-
Josh, John, and Hassan are partners in a retail lighting store. They share income and losses in the ratio of 2:2:1, respectively. The partners have agreed to liquidate the partnership. The...
-
Calculate the number of neutrons of 239Pu.
-
Give an example of a hydrocarbon that, in theory, exhibits each of the following bond angles: 60, 90, 109.5, 120, and 180.
-
What is an alkene? What is an alkyne? What are the general formulas for alkenes and alkynes, assuming one multiple bond in each? What are the bond angles in alkenes and alkynes? Describe the bonding...
-
Why can carbon form so many different compounds?
-
The record height of a man to date is 8 feet 11 inches ( 107 inches). If all men had identical body types, their weights would vary directly as the cube of their heights. Assume that all men do have...
-
The population of Leadville, CO is 2,600 people and is decreasing at a rate of 4.4% per year. Given the equation P(t) = 2600(.956), where P represents the population of Leadville and t represents the...
-
An order for a 148 lb patient is for 0.03 g/kg/hour for 2 hours bid. The medication is delivered in a 650 mg mixed in 250 ml bag of NSS. The setup is 10 gtt/mL. What is the flow rate in gtt/min?

Study smarter with the SolutionInn App


