Hydrazine (N 2 H 4 ) is used as a fuel in liquid-fueled rockets. When hydrazine reacts
Question:
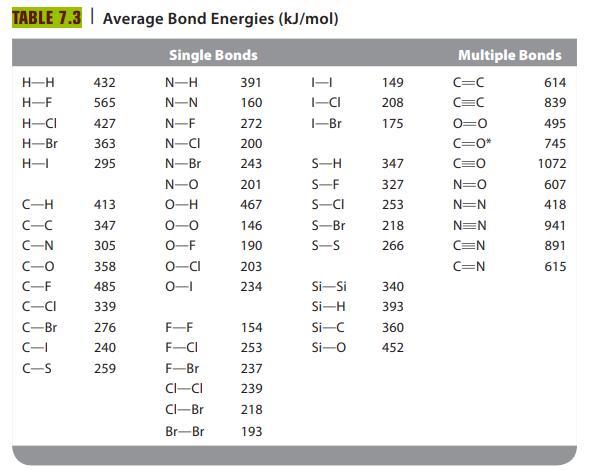
Hydrazine (N2H4) is used as a fuel in liquid-fueled rockets. When hydrazine reacts with oxygen gas, nitrogen gas and water vapor are produced. Write a balanced equation and use bond energies from Table 7.3 to estimate ΔH for this reaction.
Transcribed Image Text:
TABLE 7.3 Average Bond Energies (kJ/mol) Single Bonds N-H N-N H-H H-F H-CI H-Br H-1 C-H C-C C-N C-O C-F -CI C-Br C-I C-S 432 565 427 363 295 413 347 305 358 485 339 276 240 259 N-F N-CI N-Br N-O O-H 0-0 O-F O-CI 0-1 F-F F-CI F-Br CI-CI Cl-Br Br-Br 391 160 272 200 243 201 467 146 190 203 234 154 253 237 239 218 193 H I-CI 1-Br S-H S-F S-CI S-Bri S-S Si-Si Si-H Si-C Si-O 149 208 175 347 327 253 218 266 340 393 360 452 Multiple Bonds C=C C=C 0=0 C=O* C=0 N=O N=N N=N C=N C=N 614 839 495 745 1072 607 418 941 891 615
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The balanced chemical equation for the reaction between hydrazine N2H4 and oxygen ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write an equation for the reaction of hydrazine with fluorine gas to produce nitrogen gas and hydrogen fluoride gas. Estimate ÎH for this reaction, using bond energies from Table 13.6. Table...
-
A promising new material with great potential as a fuel in solid rocket motors is ammonium dinitramide [NH4N(NO2)2]. a. Draw Lewis structures (including resonance forms) for the dinitramide ion...
-
The steering rockets in space vehicles use N 2 O 4 and a derivative of hydrazine, 1,1-dimethylhydrazine (Study Question 5.86). This mixture is called a hypergolic fuel because it ignites when the...
-
Problem 3.6.1 Given the random variable Y in Problem 3.4.1, let U = g(Y) = Y2. (a) Find Pu(u). (b) Find Fu(u). (c) Find E[U].
-
Powderhorn Corporation reported sales of $257,000, net income of $45,300, cash of $9,300, and net cash provided by operating activities of $21,200. Accounts receivable have increased at three times...
-
Map the upper half z-plane onto the unit disk |w| < 1 so that 0, , 1 are mapped onto 1, i, i, respectively. What are the boundary conditions on |w| = 1 resulting from the potential in Prob. 9? What...
-
Nereus Montemayor was an employee of VZ Hogs, a company that raises hogs and produces hog feed. VZ Hogs used an extruder manufactured by Sebright Products, Inc. to create hog feed out of discarded...
-
The Shirt Shop had the following transactions for T-shirts for 2016, its first year of operations: During the year, The Shirt Shop sold 810 T-shirts for $30 each. Required a. Compute the amount of...
-
Knowing the specific heat of ice and water is 2.05KJ/kg o C and 4.18kJ/kgC. What is the heat needed to change 2kg of ice at -20 C into 100 C water?
-
Although nitrogen trifluoride (NF 3 ) is a thermally stable compound, nitrogen triiodide (NI 3 ) is known to be a highly explosive material. NI 3 can be synthesized according to the equation a. What...
-
Give the Lewis structure, molecular structure, and hybridization of the oxygen atom for OF 2 . Would you expect OF 2 to be a strong oxidizing agent like O 2 F 2 discussed in Exercise 61? Exercise 61....
-
Suppose we are given a range-searching data structure D that can answer rangesearching queries for a set of n points in d-dimensional space for any fixed dimension d (like 8, 10, or 20) in time that...
-
Key Features of a records management system: What are important features of the system? eg: cloud based, CRM capabilities, trust accounting.
-
what are the impacts on financial statement Potential initial increase in costs due to transition?
-
An electrical circuit board in the main control panel of a machine has four components A, B, and C connected in parallel. 300 Unites. of the circuit board were subjected to accelerated operation...
-
Measuring performance Ethan Coleman, the president and CEO of High-Tech Inc., recently stated: We are in the business of innovation! If we don't innovate and encourage creative thinking in our...
-
Using your improved business communication skills, make a thank you letter for someone who has influenced your career, your education, or your life. In writing, please use your resume header, and...
-
Beauty Island Corporation began operations on April 1 by issuing 60,000 shares of $5 par value common stock for cash at $13 per share. On April 19, it issued 2,000 shares of common stock to attorneys...
-
In Problems 718, write the augmented matrix of the given system of equations. f0.01x0.03y = 0.06 [0.13x + 0.10y = 0.20
-
VSEPR (valence state electron pair repulsion) theory was formulated to anticipate the local geometry about an atom in a molecule (see discussion in Section 25.1). All that is required is the number...
-
Chemists recognize that the cyclohexyl radical is likely to be more stable than the cyclopentylmethyl radical, because they know that six-membered rings are more stable than five-membered rings and,...
-
The presence of the carbonyl group in a molecule is easily confirmed by an intense line in the infrared spectrum around 1700 cm 1 that corresponds to a CO stretching vibration. Locate this line in...
-
Ms. Krause and Ms. Pittman were friends who had a plan to meet for coffee at Ms. Pittman's home. Unfortunately, Ms. Krause slipped and fell on icy ground on Ms. Pittman's property. As a result, Ms....
-
ces Mr. and Mrs. Udall live in a home that Mrs. Udall inherited from her parents. This year, the Udalls took out a first mortgage secured by the home. Assume the taxable year is 2023. Required: a....
-
How would you advise the HR manager to proceed in relation to the managing director?4. Are there any steps that the HR Manager should take to prevent such a situation arising again in the future? A...

Study smarter with the SolutionInn App


