Write Lewis structures for the following. Show all resonance structures where applicable. a. NO, NO3, NO4 (NO4
Question:
Write Lewis structures for the following. Show all resonance structures where applicable.
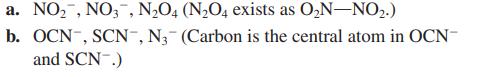
Transcribed Image Text:
a. NO₂, NO3, N₂O4 (N₂O4 exists as O₂N-NO₂.) b. OCN-, SCN-, N₂- (Carbon is the central atom in OCN- and SCN .)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a NO2 To draw the Lewis structure for NO2 first calculate the total number of valence electrons Nitr...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write Lewis structures for the following species, including all resonance forms, and show formal charges: (a) HCO2- , (b) CH2NO2- . Relative positions of the atoms are as follows: H C
-
Draw Lewis structures for the following. Show all resonance structures, where applicable. Carbon is the central atom in OCN2 and SCN2. a. NO2-, NO3-, N2O4( N2O4 exists as O2NONO2.) b. OCN2, SCN2, N3-
-
Write Lewis structures for the Kekulé representations of benzene. Show all the valence electrons.
-
Calculate the following ratios from the data given below: 1. Debt ratio 2. Debt service coverage multiples 3. Interest coverage Balance Sheet 12/31/2014 Assets Cash..$575,000 Short-term...
-
Use a program like Crystal Ball to generate Monte Carlo simulations of the profits of Safe Air and Metallwerke under various contracting clauses.
-
During every election in a particular region, pollsters conduct exit polls to help determine which candidate people voted for. During the most recent election, pollsters incorrectly predicted...
-
Juliette Shulof Furs (JSF) was a New York corporation that had been in the fur-dealing business for 15 years. George Shulof, an officer of JSF, attended two auctions conducted by Finnish Fur Sales...
-
Computing materials, labor, and cost variances The following data were drawn from the records of Inman Corporation. Planned volume for year (static budget)...... 4,000 units Standard direct materials...
-
Discuss the breath of responsibilities required of a radiographer. List the five main areas of responsibility in each specialty of radiologic technology. Discuss advantages to working in each of the...
-
Write the formula for each of the following compounds: a. Zinc chloride b. Tin(IV) fluoride c. Calcium nitride d. Aluminum sulfide e. Mercury(I) selenide f. Silver iodide
-
Write Lewis structures that obey the octet rule for each of the following molecules. a. CC1 4 b. NC1 3 c. SeCl 2 d. ICI In each case, the atom listed first is the central atom.
-
By using this sample as their own control group, these researchers were able to minimize the effect of differences among the participants and see the effect of the intervention more clearly. What did...
-
Suppose you stand with one foot on ceramic flooring and one on a wool carpet, making contact over an 78.0 cm 2 area with each foot. Both the ceramic and the carpet are 1.50 cm thick and are 10.0C on...
-
How are the increase in retained earnings and stock repurchases calculated? Sources of Funds Increase in Bank Debt Decerase in cash Decrease in account receivable Increase in retained earnings...
-
The following three stocks are available in the market: E(R) Stock A 11.1 % 1.20 Stock B 13.0 1.00 Stock C 15.5 1.40 Market 14.5 1.00 Assume the market model is valid. The return on the market is...
-
The drinking water supplied to a region of 10 million people had polychlorinated biphenyls (PCBs) level equal to the EPA's maximum contaminant level (MCL) of 0.5 g/L for PCBs. a. Based on the EPA...
-
Retro Inc. has 1,100 bonds outstanding that are selling for $992 each. The bonds carry a 6.0 percent coupon, pay interest semi-annually, and mature in 7.5 years. The company also has 9,500 shares of...
-
Chris North is the founder and president of North Enterprises, a real estate development venture. The business transactions during April while the company was being organized are listed below. Apr. 1...
-
What key concerns must functional tactics address in marketing? Finance? POM? Personnel?
-
On a hot day, a 200.0-mL sample of a saturated solution of PbI 2 was allowed to evaporate until dry. If 240 mg of solid PbI 2 was collected after evaporation was complete, calculate the K sp value...
-
The K sp of Al(OH) 3 is 2 10 -32 . At what pH will a 0.2-M Al 3+ solution begin to show precipitation of Al(OH) 3 ?
-
Calculate the equilibrium concentrations of NH 3 , Cu 2+ , Cu(NH 3 ) 2+ , Cu(NH 3 ) 2 2+ , Cu(NH 3 ) 3 2+ , and Cu(NH 3 ) 4 2+ in a solution prepared by mixing 500.0 mL of 3.00 M NH 3 with 500.0 mL...
-
1. Donna enters into an investment contract that will guarantee her 4% per year if she deposits $3,500 each year for the next 10 years. She must make the first deposit one year from today, the day...
-
what are the administrative search exception to the 4th Amendment of the constitutuion ?
-
Identify a contemporary issue concerning freedom of expression under the First Amendment, and discuss the most likely future scenarios, referencing both historical analogs and case law.?

Study smarter with the SolutionInn App


