Balance the following equations. (a) Zn(s) + VO+ (aq) Zn+ (aq) + V+ (aq) (b) Zn(s)
Question:
Balance the following equations.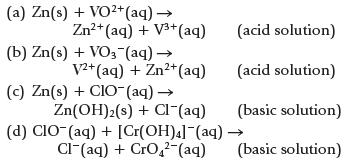
Transcribed Image Text:
(a) Zn(s) + VO²+ (aq) → Zn²+ (aq) + V³+ (aq) (b) Zn(s) + VO3(aq) → V2+ (aq) + Zn²+ (aq) (c) Zn(s) + CIO- (aq) → Zn(OH)₂(s) + Cl¯(aq) (d) CIO- (aq) + [Cr(OH)4]¯(aq) Cl(aq) + CrO2²-(aq) (acid solution) (acid solution) (basic solution) (basic solution)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
a Zns VO2aq Zn2aq V3aq To balance this equation you need to make sure the number of zinc Zn atoms va...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
Balance the following equations and indicate whether they are combination, decomposition, or combustion reactions: (a) C3H6(g) + O2 (g) CO2 (g) + H2O(g) (b) NH4NO3(s) N2O(g) + H2O(g) (c) C5H6O(I) +...
-
Bingl is one the provinces in Turkey with a very high earthquake risk, since the city center is located on the fault and is very close to the East Anatolian Fault Zone, which is also one of the most...
-
Most accounting professionals would agree that the accounting profession has developed effective tools for measuring and reporting events involving tangible assets. Most might also agree that the...
-
What are some of the benefits or uses of ratio analysis?
-
A system in which mass as well as energy can not be exchanged with the surroundings is called (a) Open system (b) Closed system (c) Isolated system (d) None of these.
-
Rembrandt Paint Company had the following income statement items for the year ended December 31, 2011 ($ in 000s): In addition, during the year the company completed the disposal of its plastics...
-
1. [20 marks] Neatly work out the following bitwise operations. Show the work step by step. Give the answers in binary, hexadecimal and decimal. a. unsigned char Question1A = (0xDB & 0x69) | 17; b....
-
Write balanced equations for the following half-reactions. (a) UO 2 + (aq) U 4 + (aq) (acid solution) (b) ClO 3 (aq) Cl (aq) (acid solution) (c) N 2 H 4 (aq) N 2 (g) (basic solution) (d) ClO ...
-
In the presence of oxgyen and acid, two half-reactions responsible for the corrosion of iron are Calculate the the standard potential, E, and decide whether the reaction is product-favored at...
-
Can you cite an example in which the acceleration of a body is opposite in direction to its velocity? If so, what example can you cite to your classmates?
-
Draw a short-run marginal cost curve, short-run average cost curve, and long-run average total cost curve for an efficient firm producing where there are diseconomies of scale.
-
Is an oligopolist more or less likely to engage in strategic decision making compared to a monopolistic competitor?
-
Why could diseconomies of scale never occur if production relationships were only technical relationships?
-
A student has just written on an exam that, in the long run, fixed cost will make the average total cost curve slope downward. Why will the professor mark it incorrect?
-
If a firm is experiencing learning by doing, what is likely true about the long-run average total cost curve? Explain your answer.
-
A fashion retailer in Santa Barbara, California, presents a new designer dress at one of the "by invitation only" fashion shows. After the show, the dress will be sold at the company's boutique store...
-
What is your assessment of the negotiations process, given what you have studied? What are your recommendations for Mr. Reed? You must justify your conclusions
-
An association has 15 voting members. Executive meetings of this association can be held only if more than half (i.e., at least eight) of the members are present (i.e., eight is the minimum quorum...
-
(a) Draw the block diagram for a divider that divides an 8-bit dividend by a 5-bit divisor to give a 3-bit quotient. The dividend register should be loaded when St = 1. (b) Draw an SM chart for the...
-
Draw an SM chart for the BCD-to-binary converter of Problem 4.13. Data from Problem 4.13. This problem involves the design of a BCD-to-binary converter. Initially a 3-digit BCD number is placed in...
-
Explain how prepayment risk affects the value of an Interest-only tranche and the Principal-only tranche in a CMO.
-
0.5 7 points Print Problem: Module 3 Textbook Problem 7 Learning Objective: 3-7 Using the straight-line method show how bonds issued at a premium affect financial statements The Square Foot Grill,...
-
A Federally qualified community health center is implementing an electronical medical record (EMR) costing $35,000 per physician provider. Identify the benefits of the EMR. Identify the ways those...

Study smarter with the SolutionInn App


