Balance the following equations involving organic compounds. (a) Ag*(aq) + CH;CHO(aq) Ag(s) + C6H5COH(aq) (b) CHCHOH +
Question:
Balance the following equations involving organic compounds.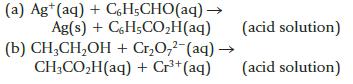
Transcribed Image Text:
(a) Ag*(aq) + CH;CHO(aq)— Ag(s) + C6H5CO₂H(aq) (b) CH₂CH₂OH + Cr₂O,2 (aq) → CH3CO₂H(aq) + Cr³+ (aq) (acid solution) (acid solution) 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
Balanced equations a Ag aq C6H5CHOaq AgC6H5O2s CH3CO2Haq acid solution b CH3CH2OH Cr2O72aq CH2CO2Haq ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Pecunious Products, Inc.'s financial results for the past three years are summarized below: Year 3 Year 2 Year 1 Sales trend 179.2 161 140 Current ratio 3.5 3.22 3.08 Acid-test ratio 1.12 1.26 1.54...
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
In the diagram, the positive terminal of the 12 V battery is grounded it is at zero potential. At what potential is point X? 12 V 4 V Ground
-
Twelve states, some local governments, and private organizations sued the EPA, contending that it did not live up to its obligation under the Clean Air Act to regulate greenhouse gases that result...
-
Evaluate the following statement: If people do not use paper or if they recycle paper, there is less incentive for lumber companies to plant trees on private land.
-
You are a store owner in downtown Terrapin. You have noticed that your business has fallen 20 percent since the city began a new, tougher approach to controlling parking in the downtown core. If...
-
Mordica Company identifies three activities in its manufacturing process: machine setups, machining, and inspections. Estimated annual overhead cost for each activity is $150,000, $325,000, and...
-
on e John Hanson starts a new busness by depositing $20, ovo bank accomf for Hanson Pest Contul. Jolm Hamsm Capital is @ Debited credited o Nof affected b 'c
-
A voltaic cell is constructed in which one half-cell consists of a silver wire in an aqueous solution of AgNO 3 . The other half-cell consists of an inert platinum wire in an aqueous solution...
-
Write balanced equations for the following reduction half-reactions involving organic compounds. (a) HCOH CHO (b) C6H5COH C6HCH3 (c) CH,CH,CHO (d) CH3OH CH CHCHCHOH (acid solution) (acid solution)...
-
Explain the trade-off between cost and accuracy in a forecasting system.
-
Assume the following information (figures are in thousands): a. Calculate the monthly budget and monthly cumulative budgets for the project. b. Draw a project S-curve representation of this project....
-
Assume that you are a project manager in a software firm and have been asked to calculate the expected cost for a new fast-food epos system. Referring to historical information, you know that the...
-
Dormant eggs of the zooplankton Daphnia survive in lake sediments for decades, making it possible to measure their physiological traits in past years. Hairston et al. (1999) extracted Daphnia eggs...
-
For each of the following events, determine the amount of freight paid by The Book Shop. Also indicate whether the freight cost would be classified as a product or period (selling and administrative)...
-
The following account titles and balances were taken from the adjusted trial balance of Scoggins Sales Co. at December 31, 2011. The company uses the periodic inventory method. Required a. Prepare a...
-
(a) Find the generating function for the sequence 0, 1, 3, 6, 10, 15, . . . (where 1, 3, 6, 10, 15, . . . are the triangular numbers of Example 4.5). (b) For n Z+, determine a formula for the sum of...
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
Quasimonochromatic light having an irradiance of 400 W/m 2 is incident normally on the cornea (n c = 1.376) of the human eye. If the person is swimming under the water (n w = 1.33), determine the...
-
Compare the amplitude reflection coefficients for an airwater (n w = 4/3) interface with that of an aircrownglass (n g = 3/2) interface, both at near-normal incidence. What are the corresponding...
-
Use Eq. (4.42) and the power series expansion of the sine function to establish that at near-normal incidence we can obtain a better approximation than the one in Problem 4.45, which is [-r ¥ ]...
-
delet question
-
Texas Petrochemical reported the following April activity for its VC-30 lubricant, which had a balance of 395 qts.@ $4.30 per quart on April 1. Purchases: Apr. 10 Apr. 14 Apr. 20 595 qts @ $4.40 495...
-
A company manufactures and sells a product for $124 per unit. The company's fixed costs are $72,760, and its variable costs are $94 per unit. The company's break-even point in units is: Multiple...

Study smarter with the SolutionInn App


