Bromine is obtained from brine wells. The process involves treating water containing bromide ion with Cl 2
Question:
Bromine is obtained from brine wells. The process involves treating water containing bromide ion with Cl2 and extracting the Br2 from the solution using an organic solvent. Write a balanced equation for the reaction of Cl2 and Br−. What are the oxidizing and reducing agents in this reaction? Using the table of standard reduction potentials (Appendix M), verify that this is a product-favored reaction at equilibrium.
Data given in Appendix M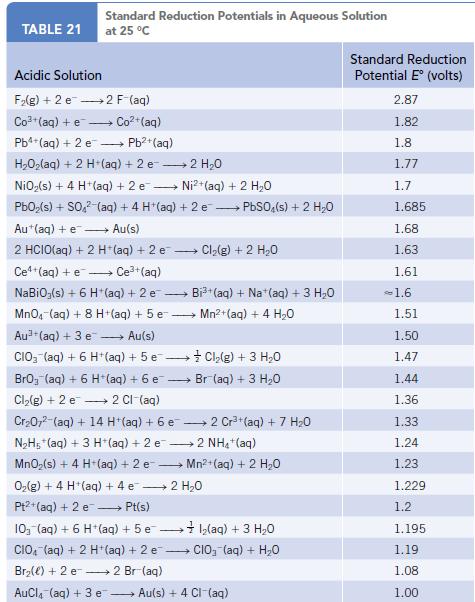
Transcribed Image Text:
Standard Reduction Potentials in Aqueous Solution TABLE 21 at 25 °C Acidic Solution F₂(g) + 2 e 2 F-(aq) Co² (aq) + e- →→Co²+ (aq) Pb²+ (aq) + 2 e - Pb²+ (aq) H,Oz(aq) + 2 H+(aq) + 2 e — 2 H,O NiO₂ (s) + 4 H+ (aq) + 2 e Ni²+ (aq) + 2 H₂O PbO₂ (s) + SO4² (aq) + 4 H*(aq) + 2 e PbSO4(s) + 2 H₂O - Au (aq) + e→→→→ Au(s) Ce (aq) + e. - 2 HCIO(aq) + 2 H+ (aq) + 2 e Ce³+ (aq) NaBiO;(s) + 6 H*(aq) + 2 e → MnO₂ (aq) + 8 H+ (aq) + 5 e Au (aq) + 3 e →→ Au(s) CIO₂ (aq) + 6 H+(aq) + 5 e- → + Cl₂(g) + 3 H₂O BrO₂ (aq) + 6 H+ (aq) + 6 e Br(aq) + 3 H₂0 → 2 Cl-(aq) Cl₂(g) + 2 e Cr₂O7²-(aq) + 14 H+ (aq) + 6 e → N₂H5+ (aq) + 3 H+ (aq) + 2 e2 NH₂ + (aq) MnO₂ (s) + 4 H+ (aq) + 2 e-- → Mn²+ (aq) + 2 H₂O → ► Cl₂(g) + 2 H₂O → Br₂(e) + 2 e→→ 2 Br (aq) - AuCl(aq) + 3 e - O₂(g) + 4 H+ (aq) + 4 e → 2 H₂O - Pt2+(aq) + 2 e→→→→ Pt(s) 10₁ (aq) + 6 H (aq) + 5 e- CIO, (aq) + 2 H+ (aq) + 2 e - Bi³+(aq) + Na (aq) + 3 H₂O Mn²+ (aq) + 4 H₂O 2 Cr³+ (aq) + 7 H₂O •Ỉ I,(aq) + 3 H,O CIO₂ (aq) + H₂O Au(s) + 4 CI-(aq) Standard Reduction Potential E° (volts) 2.87 1.82 1.8 1.77 1.7 1.685 1.68 1.63 1.61 -1.6 1.51 1.50 1.47 1.44 1.36 1.33 1.24 1.23 1.229 1.2 1.195 1.19 1.08 1.00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a Balanced chemical equation for the reaction of Cl Br Cl2g 2B ...View the full answer

Answered By

Mamba Dedan
I am a computer scientist specializing in database management, OS, networking, and software development. I have a knack for database work, Operating systems, networking, and programming, I can give you the best solution on this without any hesitation. I have a knack in software development with key skills in UML diagrams, storyboarding, code development, software testing and implementation on several platforms.
4.90+
49+ Reviews
119+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
After-tax cash flows: Year 1 2 3 4 Project Alpha R 0 37 000 72 400 246 000 Project Omega Both projects require an initial investment of R235 400 and the cost of capital is 12%. As the project manager...
-
What would you expect to happen when chlorine gas, Cl2, at 1 atm pressure is bubbled into a solution containing 1.0 M F and 1.0 M Br at 25oC? Write a balanced equation for the reaction that occurs.
-
When magnesium metal is burned in air (Figure 3.6), two products are produced. One is magnesium oxide, MgO. The other is the product of the reaction of Mg with molecular nitrogen, magnesium nitride....
-
Does the performance of kinross gold cooperation affect the performance S&P/TSX Composite index? Explain why or why not using this graph in brief Kinross Gold Corporation (K.TO) Toronto - Toronto...
-
The management of Genco Utilities Inc. is considering two capital investment projects. The estimated net cash flows from each project are as follows: The generating unit requires an investment of...
-
Match the following internal controls with the categories of control activities. Internal Controls a. Separate handling cash (receipt and deposit) func- tions from recordkeeping functions (recording...
-
Allan and Koraev both owned condominiums in the same building. Koraevs unit was directly above Allans. While Allan lived in her own unit, Koraev leased his. The leasing of Koraevs unit was managed by...
-
Jordan Bicycle Manufacturing Company currently produces the handlebars used in manufacturing its bicycles, which are high-quality racing bikes with limited sales. Jordan produces and sells only...
-
Anika who lives in Vancouver, purchased a piece of equipment from Australia for A$45,400. He was charged 19% duty, 5% GST, and 7% PST to import it. Calculate the total cost of the equipment. Assume...
-
The mineral claudetite contains the element arsenic in the form of arsenic(III) oxide, As 2 O 3 . The As 2 O 3 in a 0.562-g sample of the impure mineral was converted first to H 3 AsO 3 and then...
-
Steps have been taken to limit phosphorus is surface water. However, the amounts of nitrogen containing species in surface water can also be a problem because they also promote excessive growth of...
-
Below is the link of the video, after watching the video, Supply Chain Management: The Beer Game, write a paper about this video. https://www.youtube.com/watch?v=7ilJRuAapvA
-
A uniformly charged spherical shell has inner and outer radii of 28.0 cm and 32.0 cm, respectively. A point charge of -45.0 nC is located at the center of the spherical shell. A proton orbits around...
-
1. Write a 3,000word on family impact analysis of an issue, a specific law, a legislative proposal, or program, according to the following format. (the topic is palimony laws) Using the family...
-
Family incomes have a mean of $60 000 with a standard deviation of $20 000. The data are normally distributed. A new tax law designed for the middle class is to help families with incomes from $45...
-
How does globalization influence labor markets and employment patterns, including the phenomenon of outsourcing, offshoring, and the rise of the gig economy, and what are the social consequences of...
-
How has the process of globalization reshaped the geopolitical landscape, and what are the key drivers behind the acceleration of global interconnectedness in the 21st century?
-
At approximately 4:00 a.m. on July 14, 2000, a woman wearing a mask and pointing a firearm demanded that 7-Eleven employees at a store in Berkeley County, West Virginia, give her the store's money....
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
Identify the static 1-hazards in the following circuit. State the condition under which each hazard can occur. Draw a timing diagram (similar to Figure 1-10(b)) that shows the sequence of events when...
-
Find all of the 1-hazards in the given circuit. Indicate which changes are necessary to eliminate the hazards. c'
-
(a) Find all the static hazards in the following circuit. For each hazard, specify the values of the input variables and which variable is changing when the hazard occurs. For one of the hazards,...
-
(A) Using the above estimated regression equation for the seasonal demand for coffee in the United States, and predicting that the values of the independent or explanatory variables (P,Y and P') in...
-
Prepare the journal entry on February 5, 2025, for Crane when the wiring base is delivered to the customer. (Credit account titles are automatically indented when the amount is entered. Do not indent...
-
Current operating income for Bay Area Cycles Company is $44,000. Selling price per unit is $100, the contribution margin ratio is 20%, and fixed expense is $176,000. Required: 1. Calculate Bay Area...

Study smarter with the SolutionInn App


