Cerium, as noted in Applying Chemical Principles: 22.3 The Rare Earths, is a relatively abundant lanthanide element
Question:
Cerium, as noted in Applying Chemical Principles: 22.3 The Rare Earths, is a relatively abundant lanthanide element that has some important uses. Cerium(IV) oxide, CeO2, is widely used as a polishing agent for glass. Cerium(III) sulfide, Ce2S3, is becoming more widely used as a red pigment to replace cadmium pigments, which are environmentally less desirable.
(a) Give the electron configurations (using the noble gas notation) for Ce, Ce3+, and Ce4+.
(b) Is either Ce3+ or Ce4+ paramagnetic? If so, how many unpaired electrons does each have?
(c) The solid state structure for CeO2 is shown below. Describe the unit cell of the compound. How is this structure related to the formula?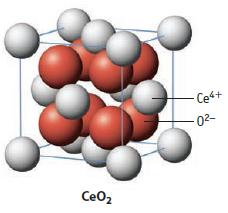
Step by Step Answer:

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel





