Consult the data in Appendix L. Are any of the nitrogen oxides listed there stable with respect
Question:
Consult the data in Appendix L. Are any of the nitrogen oxides listed there stable with respect to decomposition to N2 and O2?
Data given in Appendix L
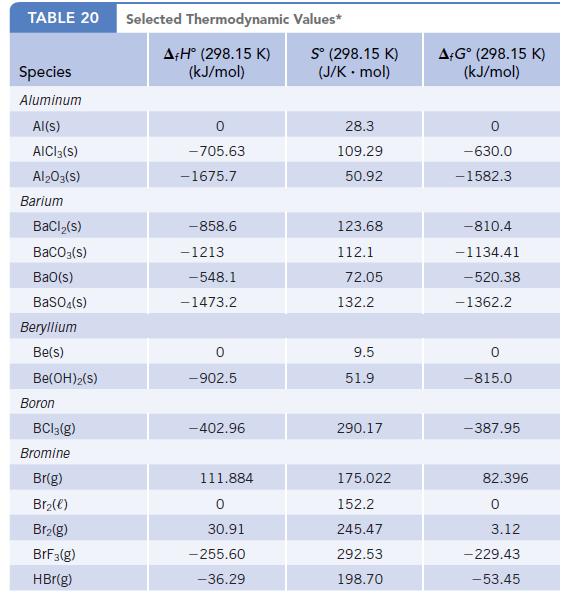
Transcribed Image Text:
TABLE 20 Species Aluminum Al(s) AICI 3(S) Al2O3(S) Barium BaCl₂(s) BaCO3(s) BaO(s) BaSO4(s) Beryllium Be(s) Be(OH)2(S) Boron BC13(g) Bromine Br(g) Br₂(e) Br₂(g) BrF3(g) HBr(g) Selected Thermodynamic A+Hº (298.15 K) (kJ/mol) 0 -705.63 -1675.7 -858.6 -1213 -548.1 -1473.2 -902.5 -402.96 111.884 0 30.91 -255.60 -36.29 Values* Sº (298.15 K) (J/K . mol) 28.3 109.29 50.92 123.68 112.1 72.05 132.2 9.5 51.9 290.17 175.022 152.2 245.47 292.53 198.70 A+Gᵒ (298.15 K) (kJ/mol) -630.0 -1582.3 -810.4 -1134.41 -520.38 -1362.2 0 -815.0 -387.95 82.396 0 3.12 - 229.43 -53.45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Consider the general decomposition reaction NO 2 N2 12 O2 F...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Explain why CsI 3 (s) is stable with respect to decomposition but NaI 3 (s) is not.
-
The stratospheric ozone (O3) layer helps to protect us from harmful ultraviolet radiation. It does so by absorbing ultraviolet light and falling apart into an O2 molecule and an oxygen atom, a...
-
The conversion of natural gas, which is mostly methane, into products that contain two or more carbon atoms, such as ethane (C2H6), is a very important industrial chemical process. In principle,...
-
Local 54 has retained James Love to represent it in a grievance against Dilated Peoples Optical Inc. The union is grieving the employers decision to exclude from the bargaining unit the position of...
-
Price regulation prevents some people from buying a good who value that good more than it costs to supply it. Apply this proposition to Figure, assuming that the state sets the price at p1 and S'...
-
The Amazon River Basin is a giant watershed with an area of about 6 million square kilometers. What does runoff in the Amazon River Basin flow toward?
-
Pressure, volume, temperature and entropy are (a) Energy properties (b) Derived properties (c) Reference properties (d) None of these.
-
Machinery purchased for $60,000 by Tom Brady Co. in 2010 was originally estimated to have a life of 8 years with a salvage value of $4,000 at the end of that time. Depreciation has been entered for 5...
-
2) Limberg Company sells TVs. The perpetual inventory was stated as $38,000 on the books at December 31, 2025. At the close of the year, a new approach for compiling inventory was used and a...
-
Which statement about ammonia is not correct? (a) Ammonia can be made by a direct reaction of the elements. (b) Aqueous solutions of ammonia are acidic. (c) Ammonia is a gas at room temperature and...
-
Construct Lewis structures for the several resonance forms of N 2 O. What is the predicted NN bond order? (a) 1 (b) 2 (c) between 2 and 3 (d) 3
-
A chemist wishes to determine the concentration of CrO42- electrochemically. A cell is constructed consisting of a saturated calomel electrode (SCE; see Exercise 24) and a silver wire coated with...
-
What are the implications for the regulatory authorities of the existence of contestable markets?
-
What is a real-life example of a monopsony?
-
Occasionally, an airline will ask passengers what the purpose of the trip is. Usually, the stated reason is to provide better service for the customer. In addition to this reason, what other...
-
How is price established in an oligopoly market?
-
What are some of the incentives for airlines to continuously improve safety?
-
Use the data and results from Exercise to prepare comparative income statements for the month of January for the company similar to those shown in Exhibit 6.8 for the four inventory methods. Assume...
-
Discrete sample spaces: suppose there are N cable cars in San Francisco, numbered sequentially from 1 to N. You see a cable car at random; it is numbered 203. You wish to estimate N. (See Goodman,...
-
Write the expression for the wavefunction of a harmonic wave of amplitude 10 3 V/m, period 2.2 10 -15 s, and speed 3 10 8 m/s. The wave is propagating in the negative x-direction and has a value of...
-
Show that if the displacement of the string in Fig. 2.12 is given by y(x, t) = A sin [kx - Ït + ε] then the hand generating the wave must be moving vertically in simple harmonic...
-
Prove that (x, t) = A cos (kx - t - /2) is equivalent to (x, t) = A sin (kx - t)
-
Assume your clinic practice has the expenses outline in the Expense Grid table (fixed, semi-variable or variable) . You have been asked to perform an analysis of cost savings opportunities. In...
-
Grand River University Water Buffalo's case Explain Recommendations with Two Points 1. Who are the stakeholders? 2. What is the ABC breakdown? How do they assign the costs ? Should they keep all...
-
You have received your bank statement, you now need to do a bank reconciliation. Every transaction must be in both the cash journal & the bank statement. Fix the discrepancies. Please check follow...

Study smarter with the SolutionInn App


