Draw the structure of glyceryl trilaurate, a fat. Lauric acid (page 941) has the formula C 11
Question:
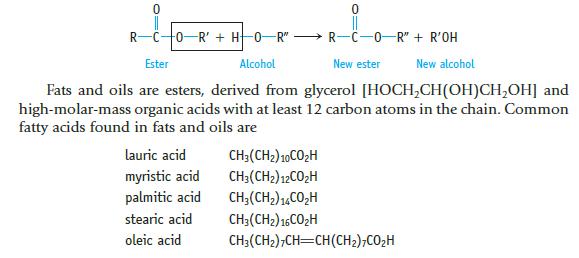
Draw the structure of glyceryl trilaurate, a fat. Lauric acid (page 941) has the formula C11H23CO2H.
(a) Write an equation for the base hydrolysis of glyceryl trilaurate.
(b) Write an equation for the reaction that could be used to prepare biodiesel fuel from this fat.
Data given in Page 941
Transcribed Image Text:
RCO R' + H0-R" - R-C-0-R" + R'OH Ester Alcohol New ester New alcohol Fats and oils are esters, derived from glycerol [HOCH₂CH(OH)CH₂OH] and high-molar-mass organic acids with at least 12 carbon atoms in the chain. Common fatty acids found in fats and oils are lauric acid myristic acid palmitic acid stearic acid oleic acid CH3(CH2) 10CO₂H CH3(CH₂) 12CO₂H CH₂(CH₂)14CO₂H CH3(CH2) 16CO₂H CH3(CH₂)7CH=CH(CH₂)7CO₂H
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
A study was conducted on a sample of 53 patients presenting with prostate cancer who had also undergone a laparotomy to ascertain the extent of nodal involvement (Collett 1991). The result of the...
-
As noted in Table 3.1, the pKa of acetone, CH3COCH3, is 19.2. In Table 3.1 (a) Draw the bond-line formula of acetone and of any other contributing resonance form. (b) Predict and draw the structure...
-
In the early days of his restaurant companys growth, McDonalds founder Ray Kroc knew that finding the right location was key. He had a keen eye for prime real estate locations. Today, the company is...
-
You have just completed your four-year degree at LLC University! Your student loans that you have accumulated while studying at LLC total $25,000. Since you have graduated, you must now begin...
-
Blakely Company estimates that total factory overhead costs will be $160,000 for the year. Direct labor hours are estimated to be 20,000. For Blakely Company (a) Determine the predetermined factory...
-
On March 1, 2019, White Company purchased $400,000 worth of inventory on credit with terms that require payment in 60 days but allow White to take a 1% discount if it pays in 10 days (often quoted as...
-
Annual household expenditures (in dollars) of a random sample of university professors: Find the range, mean, variance, and standard deviation of the sample data set. 37,224 40,964 43,724 36,188...
-
Construct a network for the aerospace launch project below and find its criticalpath. Preceding TE (weeks) Activities Activity a: Check controls b: Check propellants c: Check personnel d: Assemble...
-
4) Determine the support reactions for the system shown. There is a pin at A and a roller at B. The external force of 1000 N is applied at x = 2 meters. A F = 1000 N 4 meters B
-
Consider the reaction of cis-2-butene with H 2 (in the presence of a catalyst). (a) Draw the structure, and give the name of the reaction product. Is this reaction product chiral? (b) Draw an isomer...
-
Give the name of each compound below, and name the functional group involved. HO (a) HC-C-CH2CH2CH, H || (b) HC-CCH,CH,CH, (c) HC-C-c- CH (d) H,CCH2CH2-C-OH
-
You have been hired to correct the trial balance below that has been recorded improperly from the ledger to the trialbalance. SANDY Co. TRIAL BALANCE JANUARY 31, 20 1X Dr Cr Accounts Payable A. Sandy...
-
Question 1 . In general, what are the major objectives of internal control? What is their purpose? Question 2 . What are the risks associated with providing financial services such as those of a...
-
A person deposits $1000 in an account that yields 9% interest compounded annually. a) Set up a recurrence relation for the amount in the account at the end of n years. b) Find an explicit formula for...
-
What account balance will never change between the unadjusted trial balance and the adjusted trial balance?
-
Peter's accounts includes the following expenses. Select which TWO of the following costs are NOT allowable in calculating his tax - adjusted trading profit. ( i ) Fees incurred in the recovery of a...
-
The accountant at The Lunch Lady is using a component of its organization-wide enterprise resource planning (ERP) system to prepare a payroll tax return. describe about the component of the system...
-
The widespread use of salt on roads in Canada and the northern United States during the winter and acid precipitation throughout the year combine to cause rust on cars. Car manufacturers and other...
-
In the figure, two loudspeakers, separated by a distance of d1 = 2.63 m, are in phase. Assume the amplitudes of the sound from the speakers are approximately the same at the position of a listener,...
-
Compute points on the velocity profile from the pipe wall to the centerline of a 3/4-in Type K copper tube if the volume flow rate of water at 60F is 0.50 gal/min. Use increments of 0.05 in and...
-
Compute points on the velocity profile from the tube wall to the centerline of a plastic pipe, 125 mm OD 7.4 mm wall, if the volume flow rate of gasoline (sg = 0.68) at 25C is 3.0 L/min. Use...
-
A simple heat exchanger is made by welding one-half of a 1¾-in drawn steel tube to a flat plate as shown in Fig. 9.30. Water at 40°F flows in the enclosed space and cools the plate....
-
Answer the following questions. What are the developmental stage of adolescence. What is middle and late adolescences. What is middle adolescent stage. How many stages of development that a middle or...
-
Short Report with Memo format: Scenario: You are the general manager of the Red and White wine division of Wine Folly Inc. Your company has developed a new wine version. In taste tests, your target...
-
In a system with a high degree of concurrency, what role do locks, mutexes, and semaphores play in managing thread synchronization, and how do these mechanisms impact the scalability and reliability...

Study smarter with the SolutionInn App


