Explain why benzenesulfonic acid is a Brnsted acid. H-0: :0-S :0: benzenesulfonic acid
Question:
Explain why benzenesulfonic acid is a Brønsted acid.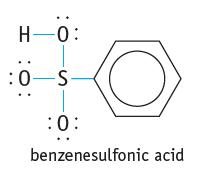
Transcribed Image Text:
H-0: :0-S :0: benzenesulfonic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Benzenesulfonic acid is a Brnsted acid because it can donate a proton H to another substa...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Work Problem 3.27 for the reverse reactions in Fig. P3.27 Problem 3.27 (a) Classify each reaction step in the forward direction with one or more of the following terms: (1) A Lewis acid-base reaction...
-
Benzenesulfonic acid is a monoprotic acid with pKa = 2.25. Calculate the pH of a buffer composed of 0.150 Mbenzenesulfonic acid and 0.125 M sodium benzenesulfonate.
-
Write a stepwise mechanism for the hydrolysis of chlorobenzene under the conditions shown in Table 24.3. TABLE 24.3 Industrial Syntheses of Phenol Reaction and comments Chemical equation Reaction of...
-
Since the early 2000s, there has been a significant increase in the price of corn-based ethanol. a. A key input in the production of corn-based ethanol is corn. Use an appropriate diagram to explain...
-
In the past decade, the Japanese economy has fallen from its lofty levels of the 1980s. As a consequence, many Japanese companies have been forced to downsize. In most companies, one of two...
-
Why is a variance analysis performed?
-
A system, consisting of \(2 \mathrm{~mol}\) of \(\mathrm{N}_{2}, 5 \mathrm{~mol}\) of \(\mathrm{H}_{2}\) and \(2 \mathrm{~mol}\) of \(\mathrm{NH}_{3}\) initially, is undergoing the following...
-
1. Is the sampling method adequate? Is the attitude-measuring scale sound? Explain. 2. Using the computerized database with a statistical software package, calculate the means of the three automotive...
-
Pressure is ratio of the amount of force being applied to the surface area over which that force is being applied. This means that you can calculate the total amount of force being applied on some...
-
m-Nitrophenol, a weak acid, can be used as a pH indicator because it is yellow at a pH above 8.6 and colorless at a pH below 6.8. If the pH of a 0.010 M solution of the compound is 3.44, calculate...
-
The chapter opening photograph (page 670) showed how the cobalt(II) chloride equilibrium responded to temperature changes. (a) Look back at that photograph. Is the conversion of the red cation to the...
-
Why do you think that for-hire logistics expenditures have quadrupled over the past thirty years in the U.S.?
-
Is an increase in the marginal income tax rate reflected by a shift in the after-tax supply of labor or a movement along the supply curve when the pretax wage rate is on the vertical axis? Explain...
-
Using the economic decision rule and opportunity cost, explain why an increase in the wage rate increases quantity of labor supplied?
-
New websites such as iFreelance.com have developed a place for companies to post projects for which freelancers can bid. What is the likely effect of this new market on market demand for freelancers?...
-
Which type of discrimination is easier to address legally demand side or institutional? Explain your answer.
-
a. List three types of demand discrimination. b. Which is the most difficult to eliminate? Why? c. Which is the easiest to eliminate? Why?
-
Fancy Paints is a small paint store. Fancy Paints stocks 200 different SKUs (stock-keeping units) and places replenishment orders weekly. The order arrives one month (let's say four weeks) later. For...
-
CLASS PERIO Solving Linear Equations: Variable on Both Sides Solve each equation. 1) 6r+ 7 = 13 + 7r 3) -7x-3x+2=-8x-8 5)-14 +66+7-26=1+5b 7) n-3n = 14-4n 2) 13-4x=1-x 4)-8-x= x - 4x 6)n+2=-14-n 8)...
-
A certain metal surface emits electrons when light is shone on it. (a) How can the number of electrons per second be increased? (b) How can the energies of the electrons be increased?
-
Would you expect the gravitational attractive force between two protons in a nucleus to counterbalance their electrical repulsion? Calculate the ratio between the electric and gravitational forces...
-
Which constituents of an atom consist of quarks and which do not?
-
Lakeside Incorporated is considering replacing old production equipment with state - of - the - art technology that will allow production cost savings of $ 1 0 , 0 0 0 per month. The new equipment...
-
If 8.86 moles of phosphorus reacts with oxygen according to the balanced chemical equation below, how many grams of oxygen are needed for a complete reaction? 4P + 5O 2 --> 2P 2 O 5
-
Given the following: US 90-day interest rate Canadian 90-day interest rate Current spot rate 90-day forward rate 4.25% 6.95% US $0.7612/C $ US $0.7782/C $ If you have $1 million and would like to...

Study smarter with the SolutionInn App


