For each of the following, tell whether the process is exothermic or endothermic. (a) HO(l) HO(s)
Question:
For each of the following, tell whether the process is exothermic or endothermic. 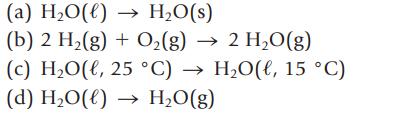
Transcribed Image Text:
(a) H₂O(l) → H₂O(s) (b) 2 H₂(g) + O₂(g) → 2 H₂O(g) (c) H₂O(l, 25 °C) → H₂O(ł, 15 °C) (d) H₂O(l) → H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Lets determine whether each of the processes is exothermic or endothermic a H2Ol H2Os ...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
For each of the following accounts, tell whether it is a permanent account or a temporary account: 1. Merchandise inventory 2. Insurance expense 3. Interest expense 4. Income taxes payable 5. Common...
-
For each of the following transactions, tell whether or not the original transaction as shown is one that results in the recognition of revenue or expenses: a. On April 15, Mikes Pressure Cleaning...
-
1) Write a generic function to integrate y(x)dx with the following format function I integrator (x, y, method) where x is a vector and y is a matrix whose columns y(:,j) are vectors of the same...
-
Explain the three types of productivity measures.
-
(a) In Figure 4.3, which box of popcorn first shows diminished marginal utility? (i) Total utility (ii) Marginal utility (b) In the cartoon "You can have too much of a good thing," which pizza slice...
-
An incompressible fluid flows outward through a blower as indicated in Fig. P5.81. The shaft torque involved, \(T_{\text {shaft }}\), is estimated with the following relationship: \[ T_{\text {shaft...
-
Kioda Corporation assembles various components used in the electronics industry. The companys major product, a computer chip, is the result of assembling three parts: X1, Y2, and Z3.The following...
-
Suppose that there is a bond with 12 years left to maturity. The face value of the bond is $1,000. Coupon rate is 15%, and YTM is 12%. Coupons are paid semiannually. What is the price that you are...
-
For each of the following, define a system and its surroundings, and give the direction of energy transfer between system and surroundings. (a) Methane burns in a gas furnace in your home. (b) Water...
-
The following terms are used extensively in thermodynamics. Define each and give an example. (a) Exothermic and endothermic (b) System and surroundings (c) Specific heat capacity (d) State function...
-
What is your feeling about the quality of products from China? In your opinion, what steps should the Chinese government take to ameliorate this problem?
-
The output of a company's mixing department during the period consists of 24000 units completed and transferred out, and 14200 units in ending Work in Process Inventory that were 25% complete as to...
-
Consider the following Bayesian network with 10 variables {X, X2,..., X10}. X3, X8( X X7 XA, Which of the following statements are true: 1. X6 X X2, X4 2. X6 X9 | X4 3. X3 X9 | X8 4. X X2 | X6 5. X4...
-
Examine the role of deconstruction in postmodern literature. What are the implications of deconstructing narrative conventions, and how does this technique alter the reader's understanding of the...
-
PLEASE HELP 1. sin cos 0 1 + sin 0 1. Given 1-sin 0 cot a. Prove the identity. (2 marks)
-
Your Pharmaceutic business (pharma lilly) needs to improve its staff performances and redesign processes and systems to address bacterial contamination with one of its medications. Do you plan to...
-
What does the balance sheet report?
-
Consider the activities undertaken by a medical clinic in your area. Required 1. Do you consider a job order cost accounting system appropriate for the clinic? 2. Identify as many factors as possible...
-
Draw bond-line structures for all constitutional isomers with molecular formula C 4 H 9 Cl.
-
(S)-2-Iodopentane undergoes racemization in a solution of sodium iodide in DMSO. Explain.
-
There are three constitutional isomers with the molecular formula C 5 H 12 . Chlorination of one of these isomers yields only one product. Identify the isomer, and draw the product of chlorination.
-
A student wishes to use their knowledge of thin lenses to make a simple projector.She wants to create a 9 3 cm tall image from a 2 . 4 cm tall slide. If the screen is 3 . 0 6 maway from the slide,...
-
Marco Company shows the following costs for three jobs worked on in April. Balances on March 31 Direct materials used (in March) Direct labor used (in March) Overhead applied (March) Costs during...
-
A car starting from rest moves in a straight line with a constant acceleration of 2 . 1 0 m / s for 1 0 . 5 s , then travels with a constant velocity for another 1 4 . 5 s . How far does it travel...

Study smarter with the SolutionInn App


