Four balloons are each filled with a different gas, each having a different density: If the density
Question:
Four balloons are each filled with a different gas, each having a different density:
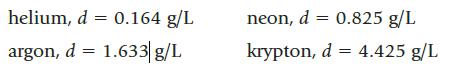
If the density of dry air is 1.12 g/L, which balloon or balloons float in air?
Transcribed Image Text:
helium, d = 0.164 g/L argon, d = 1.633 g/L neon, d = 0.825 g/L krypton, d = 4.425 g/L
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
To determine which balloons float in air we need to compare the density of each gas...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balloons are often filled with helium gas because it weighs only about one-seventh of what air weighs under identical conditions. The buoyancy force, which can be expressed as Fb =...
-
A room is filled with saturated moist air at 25oC and a total pressure of 100 kPa. If the mass of dry air in the room is 100 kg, the mass of water vapor is (a) 0.52 kg (b) 1.97 kg (c) 2.96 kg (d)...
-
A room is filled with saturated moist air at 25C and a total pressure of 100 kPa. If the mass of dry air in the room is 100 kg, the mass of water vapor is (a) 0.52 kg (b) 1.97 kg (c) 2.96 kg (d) 2.04...
-
Repeat Example 13.1, but for electrophilic substitution at C-2 or C-4 of pyridine. Explain why substitution at C-3 (eq. 13.2) is preferred. 4 H
-
Using the data in Exercise 8-13, assume that during the second year of operations O'Hara Automotive Supply Co. had net sales of $4,800,000, wrote off $114,800 of accounts as uncollectible using the...
-
An analysis of the general ledger accounts indicates that delivery equipment, which cost $200,000 and on which accumulated depreciation totaled $60,000 on the date of sale, was sold for $132,500...
-
Radiation heat transfer can occur within porous media in conjunction with conduction, as heat is radiatively transferred across pores of interstitial fluid. Under certain conditions, the effects of...
-
Sims owned property surrounded by the Morans' property. His deed was recorded in 1985, but the property had been in his family for over 50 years. He built a home in 1991. The property was accessed by...
-
At which value(s) for x does the graph of y = x-3x - 10x + 7 have horizontal tangent lines?
-
The photo below shows elemental iodine dissolving in ethanol to give a solution. Is this a physical or chemical change? Cengage Learning/Charles D. Winters Elemental iodine dissolving in ethanol.
-
Discuss the evidentiary problems raised by the following questions: a. After drinking tea made from tarragon leaves every day for a year, your grandfather reports that his bunions cleared up. Does...
-
Let X1, . . . , Xn be a random sample from the Poisson distribution with mean . Let Y = a. Prove that there is no unbiased estimator of 1/. (Write the equation that is equivalent to E (r(X)) = 1/....
-
Calculate the slope and deflection at the midpoint of the beam given in Figure. 300 N/m -3 m 3 m 600 N (B+10)x10 N.m 40 mm 200 mm C+10mm 200 mm. L C+10mm
-
Make a detailed report that Analyzes Under Amours Marketing Mix in the united states Identify Under Armour Target Group in The USA Suggest Marketing Objectives and Marketing Strategies to reach those...
-
The following is a design matrix X, response vector Y and (XX) for a multiple linear regression 3.1 Find X'Y. 1 0 1 1 02 1 0 3 X = Y = 1 1 1 1 2 52 to 1 3 (x'x) 1.33 -0.33 0.67 -0.50 0.00 -0.33 -0.50...
-
A-1 Motorcycle Sales budgets $8,000 in salaries each month. Commissions are 15% of sales, which are budgeted at $200,000 for September and October, and $280,000 for November. Salaries and commissions...
-
A bank pays interest at 6% per annum compounded every half-year. 1 How many times a year does it pay interest? 2 What percentage interest does it pay each time? 3 Complete the following table: 6%...
-
Consider the following events: 1. A petty cash fund of $200 was established on April 1, 2016. 2. Employees were reimbursed when they presented petty cash vouchers to the petty cash custodian. 3. On...
-
You work as an operations consultant for a textile company. Your client has a well-established distribution system in the US market. The company has hundreds of stores and four distribution centers....
-
Provide an IUPAC name for each of the following alcohols: a. b. c. d. e. HO. Br Br
-
Mandelate esters exhibit spasmolytic activity (they act as muscle relaxants). The nature of the alkyl group (R) greatly affects potency. Research indicates that the optimal potency is achieved when R...
-
Draw the alkoxide formed in each of the following cases: a. b. c. d. HO ? Na NaH
-
1. a) Suppose that the true linear regression model in a given situation is Y = B1+ B2X2i + Ui i = 1, 2,...,n and further that this model meets all the assumptions of Model A. Now, assume that the...
-
Springfield Limited wishes to raise 300,000 in cash. How many shares will the company have to issue to raise 300,000 if the shares have a par value of 50 cent and the shares are issued at a premium...
-
An investment of $1600 made in 2019 grew to $1800 in 2020. What is the real rate of return on this investment if the ratio of 2020 to 2019 price levels in the economy is 1.2?

Study smarter with the SolutionInn App


