Give the systematic name for each of the following alcohols, and tell if each is a primary,
Question:
Give the systematic name for each of the following alcohols, and tell if each is a primary, secondary, or tertiary alcohol:
(a) CH3CH2CH2OH
(b) CH3CH2CH2CH2OH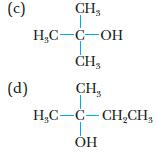
Transcribed Image Text:
(c) (d) CH₂ H₂C-C-OH CH3 CH₂ H₂C-C-CH₂CH₂ OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a CH3CH2CH2OH Systematic name 1Propanol Type of alcoh...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Give the systematic name for each of the following compounds: a. b. c. d. e. f. g. h. NH
-
Give the systematic name for each of the following compounds: a. b. CH3CH2C(CH3)3 c. CH3CH2C(CH2CH3)2CH2CH2CH3 d. e. CH3CH2C(CH2CH3)2CH(CH3)CH(CH2CH2CH3)2 f. g. h. CH3 CH3 CH,CH2CHCH2CCH3 CH3 H3...
-
Give the systematic name for each of the following compounds: a. b. c. d. e. CH CH,CHCH,CH CHCH NHCH3 CH3 CH3 CH CH-CHCH2CHCH CH3 HCH3 H2CH3 CH CHCHCH,CH CH CI CH CH2CH2CH2CHCH2CH2CH2CHa CH,CCH2CH3...
-
Clapton Company's sales budget shows the following projections for the year ending December 31, 2017: Quarters First (Jan -March) Second (April June) Third (July-Sept) Fourth (Oct-Dec) Total First...
-
Tsali Company has the following information for February: Cost of direct materials used in production .. $18,000 Direct labor ............... 54,000 Factory overhead ............ 36,000 Work in...
-
Rework Problem 55 assuming that the loan agreement calls for a principal reduction of $14,300 every year instead of equal annual payments. Data from problem 55 Prepare an amortization schedule for a...
-
The two wires are connected together at A. If the force P causes point A to be displaced horizontally 2 mm, determine the normal strain developed in each wire. C 300 mm 30 B 300 mm 30 A -P
-
The IRS has issued a summons for the tax file held by CPA Ann Whitman for her clients, the Harberts. The file consists of paper and electronic spreadsheets in which Whitman detailed some tax...
-
A firm's stock sells for $25.00 and they recently declared a dividend of $1.25 per share. Their long term growth rate is 2.5%. What is the stock's expected dividend yield for the Not yet coming year?
-
Nitration of toluene gives a mixture of two products, one with the nitro group (NO 2 ) in the ortho position and one with the nitro group in the para position. Draw structures of the two products.
-
Aromatic compounds react with a mixture of nitric and sulfuric acid to form aromatic compounds containing a nitro (NO 2 ) group. Two isomeric compounds are formed by nitration of 1,2-dimethylbenzene....
-
Positive charge Q is distributed uniformly along the x-axis from x = 0 to x = a. A positive point charge q is located on the positive x-axis at x = a + r, a distance, to the right of the end of Q...
-
You asked by USAD please provide your response not later that Monday Feb 26 "Hello As indicated in the email below, we have received a few questions from USAID concerning your budget, which were...
-
A 50-year-old professional earning $150,000 annually, is single with no dependents. He has a generous defined benefit pension plan. His registered retirement savings plan (RRSP) is worth $1 million,...
-
Ron's investment in his XYZ partnership interest on December 31, 2022, was $12,000. As the result of a serious downturn in business, the partnership calculates Ron's share of the partnership's losses...
-
What is the best strategic plan for HP Inc.'s partnership with Poly Solutions include an analysis of financial information and recommendations for increased profitability?
-
Employers who provide their employees with the use of a motor vehicle (truck, bus, etc.) for personal use, which is not included in the definition of an automobile, calculate a fair market value...
-
A paint manufacturer is attempting to improve the process that fills the 1-gallon containers. The foreperson has suggested that the nozzle can be made from several different alloys. Furthermore, the...
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
A 4-in plastic ball valve carries 600 gal/min of water at 120F. Compute the expected pressure drop across the valve.
-
A 3/4-in plastic ball valve carries 15 gal/min of water at 80F. Compute the expected pressure drop across the valve.
-
A 1-in plastic butterfly valve carries 60 gal/min of carbon tetrachloride at 77F. Compute the expected pressure drop across the valve.
-
Share details on any specific trade agreements for Egypt and research any unique business and regulatory relationships Egypt is associated with. Explain how this may help or harm the expansion of...
-
How do operating systems in a distributed environment handle resource sharing and consistency ? Explain
-
1. What is a microstate? 2. How you do determine how many microstates a system may have? 3. What does Boltzmann say about entropy?

Study smarter with the SolutionInn App


