Guanine is present in both DNA and RNA. (a) What is the most polar bond in the
Question:
Guanine is present in both DNA and RNA.
(a) What is the most polar bond in the molecule?
(b) What is the N–C=N angle in the 6-member ring?
(c) What is the N–C=N angle in the 5-member ring?
(d) What is the bond angle around the N atom of the NH2 group?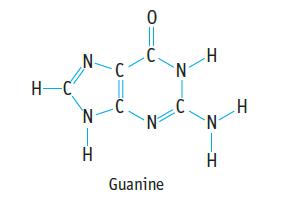
Transcribed Image Text:
H-C N- N H 0 C. N Guanine N H N H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a The most polar bond in guanine is the NH bond in the NH2 group This is because nitrogen is more el...View the full answer

Answered By

Hassan Imtiaz
The following are details of my Professional Experience. Responsibilities Eight years of demanding teaching experience in the field of finance and business studies at Master’s Level. Completion of the given tasks within given time with quality and efficiency. Marketing professional with practical experience in and solid understanding of a diverse range of management applications, including market analysis, sales and marketing, team building and quality assurance. I have excellent skills to approach deal and sustain corporate clients / customers by demonstrating not only extraordinary communication and interpersonal skills but also high caliber presentation, negotiation and closing skills. Manage and follow up the day-to-day activities. Manage and co-ordinate the inventories. Fulfillment of all the tasks assigned.
The following are details of my Areas of Effectiveness. Finance 1. Corporate Finance 2. Advanced Corporate Finance 3. Management of Financial Institutions 4. International Financial Management 5. Investments 6. Fixed Income 7. Real Estate Investment 8. Entrepreneurial Finance 9. Derivatives 10. Alternative Investments 11. Portfolio Management 12. Financial Statement Analysis And Reporting (US GAAP & IFRS) 13. International Financial Markets 14. Public Finance 15. Personal finance 16. Real estate 17. Financial Planning Quantitative Analysis 1. Time Value Of Money 2. Statistics 3. Probability Distribution 4. Business Statistics 5. Statistical Theory and Methods Economics 1. Principles of Economics 2. Economic Theory 3. Microeconomic Principles 4. Macroeconomic Principles 5. International Monetary Economics 6. Money and Banking 7. Financial Economics 8. Population Economics 9. Behavioral Economics International Business 1. Ethics 2. Business Ethics 3. An introduction to business studies 4. Organization & Management 5. Legal Environment of Business 6. Information Systems in Organizations 7. Operations Management 8. Global Business Policies 9. Industrial Organization 10. Business Strategy 11. Information Management and Technology 12. Company Structure and Organizational Management Accounting & Auditing 1. Financial Accounting 2. Managerial Accounting 3. Accounting for strategy implementation 4. Financial accounting 5. Introduction to bookkeeping and accounting Marketing 1. Marketing Management 2. Professional Development Strategies 3. Business Communications 4. Business planning 5. Commerce & Technology Human resource management 1. General Management 2. Conflict management 3. Leadership 4. Organizational Leadership 5. Supply Chain Management 6. Law 7. Corporate Strategy Creative Writing 1. Analytical Reading & Writing Other Expertise 1. Risk Management 2. Entrepreneurship 3. Management science 4. Organizational behavior 5. Project management 6. Financial Analysis, Research & Companies Valuation 7. And any kind of Excel Queries
4.80+
150+ Reviews
230+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Critically discuss functions that can be performed with project management software. What are the main differences between low-end, midrange, and high-end project management tool?
-
A weekly payroll register shows that David Wu had total wages of $397.37. He had deductions of $30.40 for FICA tax, $41.25 for federal income tax, and $17.20 for medical insurance. Compute Wu's net...
-
The molecule pictured below is epinephrine, a compound used as a bronchodilator and antiglaucoma agent. (a) Give a value for each of the indicated bond angles. (b) What are the most polar bonds in...
-
(8) (Non circular cylinder) A solid right (noncircular) cylinder has its base the R in the xy-plane and is bounded above by is 22-y + +5 5 * (x + y) dx dy paraboloid 2= x + y . The cylinder's volume....
-
What conditions and methods improve the chances that offshore outsourcing of systems development will be successful?
-
Explain both Best Practices and RBV approaches to SHRM.
-
Can earned value be used to manage a portfolio of projects? If so, how would this analysis proceed?
-
Mitakis Inc., a small service repair company, keeps its records without the help of an accountant. After much effort, an outside accountant prepared the following unadjusted trial balance as at the...
-
Suppose the government of Freedonia spends money on only defense, and gets revenue through income taxes. In 1933, the government spent $150 Million on defense and collected $110 Million worth of...
-
Methylacetamide, CH 3 CONHCH 3 , is a small molecule with an amide link (CONH), the group that binds one amino acid to another in proteins. (a) Is this molecule polar? (b) Where do you expect the...
-
Uracil is one of the bases in RNA, a close relative of DNA. (a) What are the values of the OCN and CNH angles? (b) There are two carboncarbon bonds in the molecule. Which is predicted to be shorter?...
-
What does the law of increasing costs have to do with a bowed-outward PPF?
-
The Chang reading "From pin to PIN" explores some of the differences in capitalism today from when Smith wrote The Wealth of Nations . What are some of the key differences and what are the...
-
What does a Taylor Rule do for monetary policy? Explain the concept and the importance of the Taylor principle. than Consider a simple Taylor Rule that considers only the inflation rate, it = f(t)....
-
While real interest rates drive the economy, the Fed's policy instrument is a nominal interest rate. Why don't they simply use the real rate as their policy instrument?
-
Explain why an upward-sloping yield curve implies according to the Expectations Hypothesis.
-
The optimal objective function value in a linear programming model for a profit maximization problem is $100. A non-binding constraint in this LP model is relaxed. What is the maximum attainable...
-
Using the results of the experiment described in Exercise 63, does it matter whether one listens to rap music while studying, or is it better to study without music at all? a) Test an appropriate...
-
The figure shows a bolted lap joint that uses SAE grade 8 bolts. The members are made of cold-drawn AISI 1040 steel. Find the safe tensile shear load F that can be applied to this connection if the...
-
If you flip a coin, what prediction can you make about it coming up heads in a single event?
-
If you flip a coin 1000 times, what prediction can you make about the number of times it comes up heads?
-
A superposition wave function can be expanded in the eigenfunctions of the operator corresponding to an observable to be measured. In analogy to rolling a single die, each of the infinite number of...
-
Solve the following integrals, using the most appropriate technique, integration by parts or tabulation 1. fxlnx dx 2. S cos x dx 3. (t+2+3+t-t+5)e dt 4. sen cos0d0 5. S sen0 cos/30 de 6. sec* 3x...
-
On January 1, 2022, Cullumber Corporation had the following stockholders' equity accounts. Common Stock ($26 par value, 58,500 shares issued and outstanding) Paid-in Capital in Excess of Par-Common...
-
Let f(x) = +2 +9. 1. Find f'(x) = 2. Find the critical numbers for f. Enter your answers as a list of comma separated numbers (for example: 2, -4/3) 3. By hand, find the intervals where f is...

Study smarter with the SolutionInn App


