Identify and name the water-insoluble product in each reaction and write the net ionic equation: (a) CuCl(aq)
Question:
Identify and name the water-insoluble product in each reaction and write the net ionic equation: 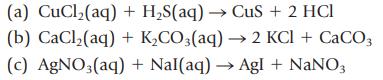
Transcribed Image Text:
(a) CuCl₂(aq) + H₂S(aq) → CuS + 2 HCI (b) CaCl₂(aq) + K₂CO3(aq) → 2 KCl + CaCO3 (c) AgNO3(aq) + Nal(aq) → Agl + NaNO3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
To identify the waterinsoluble product in each reaction and write the net ionic equation you need to ...View the full answer

Answered By

Loise Ndungu
I have five years of experience as a writer. As I embark on writing your papers from the prologue to the epilogue, my enthusiasm is driven by the importance of producing a quality product. I put premium product delivery as my top priority, as this is what my clients are seeking and what makes me different from other writers. My goal is to craft a masterpiece each time I embark on a freelance work task! I'm a freelance writer who provides his customers with outstanding and remarkable custom writings on various subjects. Let's work together for perfect grades.
4.90+
78+ Reviews
224+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Dracaris Inc. purchased an investment and expected to earn: Year 1 2 3 TV Net Cash Flows Php250 Php300 Php450 Php900 Assuming a 10% discount factor, how much is the net present value based on the...
-
1) Write balanced net ionic equation for the neutralization ofequal molar amounts of HNO2 and KOH 2) Write balanced net ionic equation for the neutralization ofequal molar amounts of HBr and NH3 3)...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
If the Federal Government increases taxes:What will be the effect on money demand, money supply, and interest rates? Money demand decreases, money supply is unchanged, and interest rates decrease...
-
In the late 1970s, Britain seemed to have struck it rich. Having developed its North Sea oil-producing fields in earlier years, Britain suddenly found its real income higher as a result of a dramatic...
-
Interpret each of the following probability statements, using the frequentist interpretation of probability. a. The probability of being dealt a pocket pair in Texas hold'em is 0.059. b. If a...
-
During a trial on single cylinder four stroke IC engine, the following observations were taken : Mean effective pressure \(=4\) bar, speed \(=200\) RPM, Brake power \(=7.5 \mathrm{~kW}, \mathrm{~L} /...
-
Coca-Cola Company is a global soft-drink beverage company (ticker symbol + KO) that is a primary and direct competitor with PepsiCo. The data in Chapter 12s Exhibits 12.1312.15 include the actual...
-
2- You own a shoe store and are deciding which shoes to stock for the upcoming season. You can either stock designer shoes, midrange shoes, or inexpensive shoes. If you stock designer shoes, you can...
-
Bromine is obtained from sea water by the following redox reaction (a) What has been oxidized? What has been reduced? (b) Identify the oxidizing and reducing agents. Cl(g) + 2 NaBr(aq) 2 NaCl(aq) +...
-
Name two anions that combine with Al 3+ ion to produce water-soluble compounds.
-
For the following exercises, graph the inequality. 1/4 x 2 + y 2 < 4
-
discuss the role of ethical leadership in building resilience and guiding organizations through times of crisis or uncertainty, by prioritizing ethical considerations and maintaining a steadfast...
-
Pharoah Industries produces and sells a cell phone-operated home security control systems. Information regarding the costs and sales during May 2022 is as follows. Unit selling price $45.00 Unit...
-
Identify the type of fund in which each activity is recorded: 1 ) The city receives a grant from the federal government to institute a meal delivery program for senior citizens. 2 ) To remedy a...
-
1) Using the information bellow calculate the operating income for 2023. Beginning finished goods, 1/1/2023 $50,000 Ending finished goods, 12/31/2023 $35,000 Beginning work in process, 1/1/2023...
-
Would you consider this social media post hateful? Note: this is just an image, not a video. Please describe why this social media post may or may not be hateful. 2. Would you consider this social...
-
The December 31, 2012, trial balances of Pettie Corporation and its 90%-owned subsidiary Sunco Corporation are as follows: Petties investment in Sunco was purchased for $1,260,000 in cash on January...
-
Discuss whether responsible human resources management should apply different standards for the home company and suppliers, for developed countries and developing countries, and for large companies...
-
Consider the oxidation of the amino acid glycine NH 2 CH 2 COOH to produce water, carbon dioxide, and urea NH 2 CONH 2 : NH 2 CH 2 COOH(s) + 3O 2 (g) NH 2 CONH 2 (s) + 3CO 2 (g) + 3H 2 O(l)...
-
Show what reagents you would use to prepare each of the following ethers via a Williamson ether synthesis, and explain your reasoning. a. b. c. OMe
-
Calculate the average CH bond enthalpy in methane using the data tables. Calculate the percent error in equating the average CH bond energy in Table 4.3 with the bond enthalpy. Table 4.3 Selected...
-
Forensic psychology is an attempt to reinvent itself and lacks originality. Discuss the value of forensic psychology to the criminal justice system.?
-
You (and your team) are employed as Management Accountants working for Qantas Airways. You have been given the task of recommending a new business strategy for Qantas Airways. Question 1 1. Using...
-
Identify proposed federal or state legislation (bill) and write a short summary (no more than 2 pages in length) of the bill. Include the following information in the bill summary: Bill number (check...

Study smarter with the SolutionInn App


