Liquid HCN is dangerously unstable with respect to trimer formationthat is, formation of (HCN) 3 with a
Question:
Liquid HCN is dangerously unstable with respect to trimer formation—that is, formation of (HCN)3 with a cyclic structure.
(a) Propose a structure for this cyclic trimer.
(b) Estimate the energy of the trimerization reaction using bond dissociation enthalpies (Table 8.8).
Data given in Table 8.8
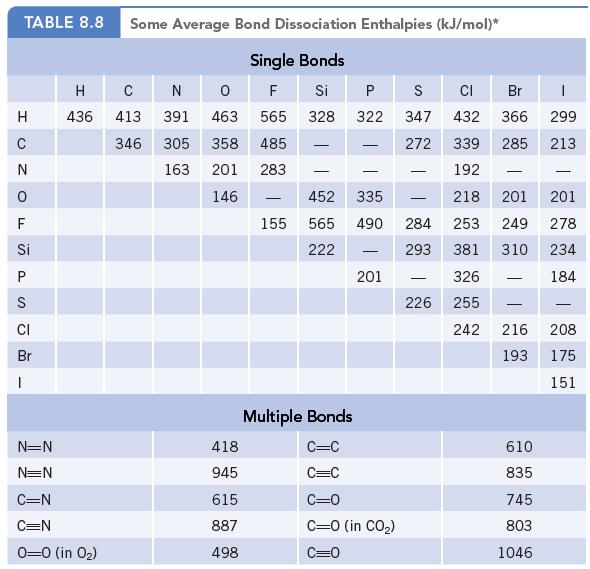
Transcribed Image Text:
TABLE 8.8 H C ZO N O FS Si P S CI Br I N=N N=N Some Average Bond Dissociation Enthalpies (kJ/mol)* Single Bonds 0 F Si 463 565 328 358 485 201 283 146 H C 436 413 346 C=N C=N 0-0 (in 0₂) N 391 305 163 418 945 615 887 498 155 - P S Multiple Bonds C=C C=C 322 - 452 335 565 490 222 201 CI Br 432 366 339 285 192 218 201 284 253 249 293 381 310 326 226 255 242 C=0 C=0 (in CO₂) C=0 347 272 - - - 1 299 213 610 835 745 803 1046 - 201 278 234 184 - 216 208 193 175 151
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Proposed structure for the cyclic trimer of HCN The cyclic trimer of HCN has a hexagonal ring stru...View the full answer

Answered By

Shivam Bhatra
A Third Year undergraduate student at Lovely Professional University with an interest in web development , Big data and Digital marketing . Seeking an internship to associate with an organization,which offers a perfect environment for growth.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
QUESTION 20 An equity analyst wants to verify whether there is a variation in the mean rate of return for 8 types of equity. The analyst selected a sample of 142 and conducted an ANOVA test that is...
-
There are four diastereomers (A D, margin) of (4S)-2-bromo-4-phenylcyclohexanol. As a team, formulate their structures and draw each diastereomer in the most stable chair conformation (see Table...
-
The lower 10-15 km of the atmosphere-the troposphere-is often in a convective steady state at constant entropy, not constant temperature. In such equilibrium pVy is independent of altitude, where =...
-
Name each of the following compounds using R,S and E,Z (Section 3.5) designations where necessary: a. b. c. d. e. f. g. h. CH2CH3 H- CH3 H2CH C-C C-C CI Br CH,CH CH-CH,CH,CH, - H3C CH2CH2l CH2CH...
-
Find the Web page for Western Digital Corporation in Santa Clara, California. a. What products does this company make? b. Using the information at www.nwasoft.com/press/mag_westdig.htm (last accessed...
-
The following information relates to Karen Weigel Co. for the year 2020. Instructions After analyzing the data, prepare (a) An income statement (b) A comprehensive income statement for the year 2020....
-
8 C 3 Perform the indicated calculation.
-
In a thin-slab, continuous casting process, molten steel leaves a mold with a thin solid shell, and the molten material solidifies as the slab is quenched by water jets en route to a section of...
-
An empty 150g glass bottle is at room temperature, 22 Celcius, if 270g of boiling water is poured into the bottle, what is the temperature of the bottle and the water when they reach thermal...
-
Use f H data in Appendix L to calculate the enthalpy change of the reaction Speculate on whether such a reaction could be used to fix nitrogen. Would research to find ways to accomplish this...
-
Calcium fluoride can be used in the fluoridation of municipal water supplies. If you want to achieve a fluoride ion concentration of 2.0 10 5 M, what mass of CaF 2 must you use for 1.0 10 6 L of...
-
If 0.654 g of zinc metal reacts with 0.321 g of yellow powdered sulfur, what is the mass of the zinc sulfide produced?
-
Suppose a pharmaceutical company produces a new drug that cures baldness. The inverse demand curve for the drug is P = 205 20Q, where Q measures the number of pills in millions. The various...
-
4 Yale students are going on a trip abroad where they will have to live close to each other. At the destination, there is a disease that spreads easily among those who live close to each other. The...
-
Border Searches of Your Electronic Devices As a disclaimer, this assignment deviates from the normal material for this chapter. However, given its relevancy and how it directly impacts anybody...
-
PU = 240 4QU and PR = 200 2QR where C = 1000 + 40Q Calculate the profit-maximizing prices and quantities for the urban and rural markets
-
Step #1: Brainstorm your purpose Purpose = talents + passions + skills/expertise + values Identify at least 4 attributes for each area of your personal and professional talents, passions,...
-
Can we infer from the data that the proportion of Americans earning at least $75,000 is greater in 2008 than in 2006 (INCOME06)? For each of the following variables, conduct a test to determine...
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Use Fig. P.5.3 to show that if a point source is placed at the focus F 1 of the ellipsoid, plane waves will emerge from the far side. Remember that the defining requirement for an ellipse is that the...
-
Construct a Cartesian oval such that the conjugate points will be separated by 11 cm when the object is 5 cm from the vertex. If n 1 = 1 and n 2 = 3/2, draw several points on the required surface
-
Experiments have shown that riders in a car begin to feel uncomfortable while traveling around a turn if their acceleration is greater than about 0.40 g. Use this fact to calculate the minimum...
-
Identify and explain the assumptions of Groupthink presented by the authors. Do you agree with 't Hart's criticism that Janis's suggestions for avoiding Groupthink "inadvertently erode collegiality...
-
1. Describe the various types of symbols that can be used to communicate an organization's values. Provide an example of each. Why are symbols important to examine when taking a cultural approach to...
-
Effective Human Capital Management is essential in every organization wishing to achieve and maintain peak performance. The days of viewing people as a cost should be phasing out completely in every...

Study smarter with the SolutionInn App


