Nitinol, a nickel-titanium alloy, is used in eyeglass frames or orthodontics. If you bend a pair of
Question:
Nitinol, a nickel-titanium alloy, is used in eyeglass frames or orthodontics. If you bend a pair of eyeglass frames out of shape, they snap back to the proper fit. That is the reason nitinol is often called “memory metal.”
Memory metal is an alloy with roughly the same number of Ni and Ti atoms. When the atoms are arranged in the highly symmetrical austenite phase, the alloy is relatively rigid. In this phase a specific shape is established that will be “remembered.” If the alloy is cooled below its “phase transition temperature,” it enters a less symmetrical but flexible phase (martensite). Below this transition temperature, the metal is fairly soft and may be bent and twisted out of shape. When warmed above the phase transition temperature, nitinol returns to its original shape. The temperature at which the change in shape occurs varies with small differences in the nickel-to-titanium ratio.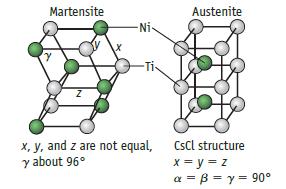
(a) What are the dimensions of the austenite unit cell? Assume the Ti and Ni atoms are just touching along the unit cell diagonal. (Atom radii: Ti = 145 pm; Ni = 125 pm.)
(b) Calculate the density of nitinol based on the austenite unit cell parameters. Does the calculated density of the austenite unit cell agree with the reported density of 6.5 g/cm3?
(c) Are Ti and Ni atoms paramagnetic or diamagnetic?
Step by Step Answer:

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel





