Nitrogen monoxide, a gas recently found to be involved in a wide range of biological processes, reacts
Question:
Nitrogen monoxide, a gas recently found to be involved in a wide range of biological processes, reacts with oxygen to give brown NO2 gas.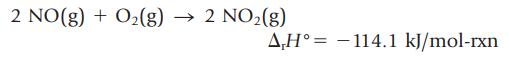
Is this reaction endothermic or exothermic? What is the enthalpy change if 1.25 g of NO is converted completely to NO2?
Transcribed Image Text:
2 NO(g) + O₂(g) →>>> 2 NO₂(g) A,H114.1 kJ/mol-rxn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The given reaction is 2 NOg O2g 2 NO2g H 1141 kJmolrxn To determine whether this reaction is endothe...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Nitrogen monoxide, a gas recently found to be involved in a wide range of biological processes, reacts with oxygen to give brown NO 2 (g) 2NO (g) + O 2 (g) 2 NO 2 (g) H = -114.1 kJ/mol-rxn Is this...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The task in this design project is to design an actuation system to power the ram's reciprocating motion in a small-size shaper. The power source of the actuation system is an AC motor with 0.75 hp...
-
Describe the alternative workplace approach.
-
The exercises below focus on the logic used in this chapter rather than on number crunching. You should try to solve them without using any of the analytical solutions from the text. Suppose that...
-
Air is delivered through a constant-diameter duct by a fan. The air is inviscid, so the fluid velocity profile is "flat" across each cross section. During the fan start-up, the following velocities...
-
The following are a number of scenarios that might constitute a violation of the Code of Professional Conduct. Required For each of the following situations, identify whether it involves a violation...
-
What are the challenges in integrating process simulation with real-time process control and automation? How can digital twins and cyber-physical systems support the seamless integration of...
-
Nitrogen gas is confined in a cylinder with a movable piston under a constant pressure of 9.95 10 4 Pa. When 695 J of energy in the form of heat is transferred from the gas to the surroundings, its...
-
A volume of 1.50 L of argon gas is confined in a cylinder with a movable piston under a constant pressure of 1.22 10 5 Pa. When 1.25 kJ of energy in the form of heat is transferred from the...
-
You can write any program using only sequence structures.
-
Mt. Hood Meadows wants to add a welcome sign to the new highway to let drivers know they are near the entrance to the resort. The sign will be placed over an equal tangent sag curve, directly over...
-
Substitute the above demand functions into the utility function to get what is known as the " indirect " utility function, which gives utility as a function of income, prices, and amenities. (b)...
-
Define a Lease and what 3 aspects need to be considered before a contract can be considered to be a lease. QUESTION 2 Provide a journal of the lease expense for the period ended 3 1 December 2 0 2...
-
Indicate whether the income amounts described would be subject to Canadian taxation. Your answer should explain whether the person is a Canadian resident. Explain the basis for your conclusions....
-
Summarize the debate between "Discretion" and "Rules" for the Federal Reserve. What is the general effect of easy monetary policy on the behavior of banks, lenders, consumers, and fraudsters (think...
-
What is the weighted- average method for process costing systems?
-
Discuss the information available from the following techniques in the analysis of inorganic pigments used in antique oil paintings: (i) Powder X-ray diffraction, (ii) Infrared and Raman...
-
For each pair of compounds, identify whether they are constitutional isomers or two representations of the same compound: (a) (b) (c) (d)
-
The following table indicates the number of constitutional isomers with molecular formula C 7 H 16 . Draw each of the isomers, making sure not to draw the same compound twice. NUMBER OF...
-
In each case below, draw a Newman projection as viewed from the angle indicated: (a) (b) (c) (d) (e) (f) Observer CI Observer CI
-
A desktop support technician recently installed two new hard drive disks (HDDs) into an office workstation, and the HDDs will represent the D: and E: drive in the Windows operating system (OS)....
-
1. Java, one of the most famous programming dialects being used today, was created by Sun Microsystems, which was procured by Oracle in 2010. Prophet had recently looked for $9 billion in penalties...
-
Question 4 (P2P Tit-for-Tat, 16%). In this question, we consider a slightly modified Tit-for-Tat algorithm in a P2P system. As shown in the figure below, A and B are communicating with their top-6...

Study smarter with the SolutionInn App


