Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation.
Question:
Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation.
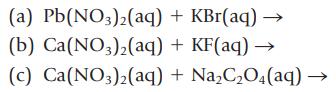
Transcribed Image Text:
(a) Pb(NO3)2(aq) (b) Ca(NO3)2(aq) (c) Ca(NO3)2(aq) + KBr(aq) → + KF(aq) → + Na₂C₂O4 (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
To predict the products of each precipitation reaction youll need to identify whether an insoluble c...View the full answer

Answered By

Gabriela Rosalía Castro
I have worked with very different types of students, from little kids to bussines men and women. I have thaught at universities, schools, but mostly in private sessions for specialized purpuses. Sometimes I tutored kids that needed help with their classes at school, some others were high school or college students that needed to prepare for an exam to study abroud. Currently I'm teaching bussiness English for people in bussiness positions that want to improve their skills, and preparing and ex-student to pass a standarized test to study in the UK.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation. (a) NiCl(aq) + (NH4)S(aq) (b) Mn(NO3)2(aq) + Na3PO4(aq)
-
Predict the products of each acid-base combination listed. Assume that a neutralization reaction occurs. a. HCl and KOH b. H2SO4 and KOH c. H3PO4 and Ni(OH)2
-
Predict the products of each acid-base combination listed. Assume that a neutralization reaction occurs. a. HBr and Fe(OH)3 b. HNO2 and Al(OH)3 c. HClO3 and Mg(OH)2
-
Ballard Ltd makes three products A, B and C. Each passes throughtwo departments: Machining and Assembly. Budgeted production ineach department by each productUnitsMachiningAssemblyPr 2 answers
-
The fundamental problem with any attempt to limit climate change is that the countries whose growth poses the greatest threat to the planet are also the countries that can least afford to pay the...
-
How are unexpected costs such as liquidation expenses, disposal costs, or unrecorded liabilities covered in the safe distribution schedule?
-
We continue our study of term life insurance demand from Chapters 3 and 4. Specifically, we examine the 2004 Survey of Consumer Finances (SCF), a nationally representative sample that contains...
-
Determining the proper costing system Professor Julia Silverman received the following e-mail message. I dont know if you remember me. I am Tim Wallace. I was in your introductory accounting class a...
-
Fairfield Company's raw materials inventory transactions for the most recent month are summarized here: Note: Assume, purchase of raw materials is on account. Beginning raw materials Purchases of raw...
-
Oxalic acid, H 2 C 2 O 4 , which is found in certain plants, can provide two hydronium ions in water. Write balanced equations (like those for sulfuric acid on page 142) to show how oxalic acid can...
-
Decide whether each of the following is watersoluble. If soluble, tell what ions are produced when the compound dissolves in water. (a) NiCl 2 (b) Cr(NO 3 ) 3 (c) Pb(NO 3 ) 2 (d) BaSO 4
-
1. Do professional standards allow a company's auditors also to provide tax services and retain their independence? 2. How have provisions of the Sarbanes-Oxley act limited a public company choice of...
-
Suppose there are three possible states of the economy and estimates of the rates of return for Stocks A and B in each state are given in the table below. 3. (12 points) Suppose there are three...
-
The solid cylinder has a radius of R = 0.020 meters and a mass of M = 0.113 kg. The cylinder is initially at rest against a spring with constant k = 143 N/m and an initial compression of x = 0.084...
-
Niki, Jeanette, and Allen are playing a three-way tug-of-war. Three ropes of equal length are tied together. Niki pulls with a force of 210 N, Jeanette pulls with a force of 190 N, and Allen pulls...
-
1. Rhea received the following income in 2021: Business income, Philippines Business income, US P 300,000 250,000 Expenses, Philippines 200,000 Expenses, US 125,000 Interest on deposit with Metrobank...
-
Taking your business online has the benefit of removing the limitation of geography: True or false
-
Does the asking price of a new car vary according to whether the dealership is in a small town, a city, or a suburban area? To test this question, a researcher randomly selects dealerships selling...
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
Predict the major product(s) for each reaction below. (a) (b) (c) 1) LAH 2) , 1) PhMgBr 2) H,0
-
Starting with cyclopentanone and using any other reagents of your choosing, identify how you would prepare each of the following compounds: (a) (b) (c) (d)
-
Predict whether the following compound will be aromatic, nonaromatic, or antiaromatic. Explain your reasoning.
-
Yukon Bike Corp. manufactures mountain bikes and distributes them through retail outlets in Canada, Montana, Idaho, Oregon, and Washington. Yukon Bike Corp. declared the following annual dividends...
-
Bremen Fitness Products produces a sports drink. On October 1, it had no work-in-process inventory. It started production of 9,400 cases of the drink in October and shipped 8,000 cases to retailers....
-
The Slumber Store (TSS) is a national wholesaler of beds, mattresses, pillows, and sheets. Key totals from TSS's most recent and forecasted financial statements are presented in the table below. From...

Study smarter with the SolutionInn App


