The products formed in several reactions are given below. Identify the reactants (labeled x and y) and
Question:
The products formed in several reactions are given below. Identify the reactants (labeled x and y) and write the complete balanced equation for each reaction. 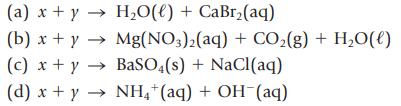
Transcribed Image Text:
(a) x + y → H₂O(l) + CaBr₂(aq) (b) x + y → Mg(NO3)2(aq) + CO₂(g) + H₂O(l) (c) x + y BaSO4(s) + NaCl(aq) → (d) x + y → NH4+ (aq) + OH(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a x y H2Ol CaBr2aq Reactants 2 HClaq CaCO3s Balanced Equation 2 HClaq CaCO3s H2Ol C...View the full answer

Answered By

Muhammad Mahtab
everyone looks that their work be perfect. I have more than a five year experience as a lecture in reputable institution, national and international. I provide perfect solution in marketing, case study, finance problems, blog writing, article writing, business plans, strategic management, human resource, operation management, power point presentation and lot of clients need. Here is right mentor who help clients in their multi-disciplinary needs.
5.00+
3+ Reviews
14+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The products formed in several reactions are given below. Identify the reactants (labeled x and y) and write the complete balanced equation for each reaction. (a) x + y (NH4)2SO4(aq) (b) x + y...
-
Write a balanced equation for each of the following reactions or reaction sequences. (a) The reaction catalyzed by PFK-2 (b) The conversion of 2 moles of oxaloacetate to glucose (c) The conversion of...
-
Write a balanced equation for each reaction. (a) (b) (c) (d) H SO, heat CH3 CH2CH-CH NaOC(CH3 3 Br Br Nal CHCH CH-CH acetone NaOH, heat CH3 CH CCH3 Br
-
Jeremiah Wedgewood, the CFO, is adamant that the company needs to move ahead with the new division. While Josey also thinks that creating a new division with a new product line is a good idea, she is...
-
How might a zero interest rate complicate the task of monetary policy?
-
In 2010, the Internal Revenue Service (IRS) sampled 308,946 tax returns to obtain estimates of various parameters. Data were published in Statistics of Income, Individual Income Tax Returns....
-
A four cylinder, four stroke cycle petrol engine develops \(40 \mathrm{~kW}\) at \(2500 \mathrm{rpm}\). The mean effective pressure is \(0.8 \mathrm{MPa}\) and mechanical efficiency is \(80 \%\)....
-
Hugh Leach Corp., a producer of machine tools, wants to move to a larger site. Two alternative locations have been identified: Bonham and McKinney. Bonham would have fixed costs of $800,000 per year...
-
What is its unit price? Let's think of a batch of 100 baked goods...what is its total price? Speculate how much is the cost to bake (AND sell) that 1 batch (100 units)? Calculate its related gross...
-
Indicate which of the following copper(II) salts are soluble in water and which are insoluble: Cu(NO3)2, CUCO3, Cu3(PO4)2, CuCl.
-
Complete and balance the equations below, and classify them as precipitation, acidbase, gasforming, or oxidationreduction reactions. Show states for reactants and products (s, , g, aq). (a) NiCO3 +...
-
(a) Describe what occurs in the process of transcription. (b) Describe what occurs in the process of translation.
-
how do you know which equation to use for sustainable growth? There are several: g*=PRAT g*= R x ROEbop g* = change in equity / equity bop g*= R x Earnings / Equity What is the formula for Actual...
-
What are the implications of incorporating machine learning algorithms, such as deep learning neural networks, in the identification and mitigation of operational risks within large-scale financial...
-
How can I overcome the challenges of measuring marketing effectiveness?
-
How do advanced risk aggregation techniques, such as nested simulation or extreme value theory, address the challenges posed by non-linear interactions and tail dependencies in multi-asset portfolios?
-
Discuss the implications of the efficient market hypothesis on the effectiveness of technical and fundamental analysis in stock valuation.
-
Apple Contractors, an 80%-owned subsidiary, is constructing a warehouse for its parent, Plum Corporation. The following information is available on December 31, 2011: Percent of completion . . . . ....
-
DC has unused FTC carryover from 2017 in the separate category for GC income as the result of income generated by a foreign branch. The income was foreign source general category income. In 2018 the...
-
Assign a name for each of the following compounds. a. b. c.
-
21.05 g of steam at 373 K is added to 415 g of H 2 O(l) at 298 K at a constant pressure of 1 bar. Is the final state of the system steam or liquid water? Calculate S for the process.
-
A van der Waals gas has a value of z = 1.00061 at 410. K and 1 bar and the Boyle temperature of the gas is 195 K. Because the density is low, you can calculate V m from the ideal gas law. Use this...
-
What does the textbook, Introduction to Forensic Psychology recommend regarding a specialized degree in forensic psychology?
-
es On October 31, 2024, Crowe Company's general ledger shows a cash account balance of $8,454. The company's cash receipts for the month total $74,700, of which $71,390 has been deposited in the...
-
On September 25, 2023, Homeowner met with Contractor to discuss an addition that Homeowner wanted to add to his house. Homeowner wanted Contractor to build a large primary bedroom with a large,...

Study smarter with the SolutionInn App


