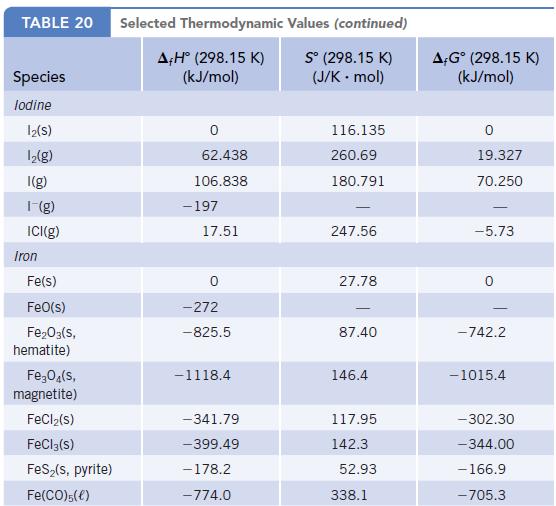
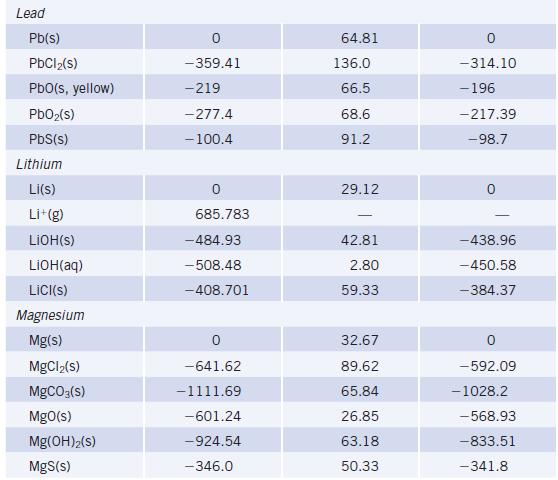
Using data in Appendix L, calculate r G values for the decomposition of MCO 3 to
Question:
Using data in Appendix L, calculate ΔrG° values for the decomposition of MCO3 to MO and CO2 where M = Mg, Ca, Ba. What is the relative tendency of these carbonates to decompose?
Data given in Appendix L
Transcribed Image Text:
TABLE 20 Species lodine 12(S) 1₂(g) I(g) 1-(g) ICI(g) Iron Fe(s) FeO(s) Fe₂O3(s, hematite) Fe₂O4(s, magnetite) FeCl₂(s) FeCl3(s) FeS₂(s, pyrite) Fe(CO)5(€) Selected Thermodynamic A,Hº (298.15 K) (kJ/mol) 0 62.438 106.838 -197 17.51 -272 -825.5 -1118.4 -341.79 -399.49 -178.2 -774.0 Values (continued) Sº (298.15 K) (J/K.mol) 116.135 260.69 180.791 247.56 27.78 87.40 146.4 117.95 142.3 52.93 338.1 A-Gº (298.15 K) (kJ/mol) 19.327 70.250 -5.73 -742.2 -1015.4 -302.30 -344.00 - 166.9 -705.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Using data in Appendix C, calculate ÎH°, ÎS°, and ÎG° at 298 K for each of the following reactions. In each case show that ÎG° = ÎH° -...
-
The decomposition of aqueous hydrogen peroxide in a given concentration of catalyst yielded the following data: Verify that the reaction is first order. Determine the rate constant for the...
-
Using data in Appendix E, calculate the standard emf for each of the following reactions: (a) H2(g) + F2(g) 2 H+ (aq) + 2 F- (aq) (b) Cu2+(aq) + Ca(s) Cu(s) + Ca2+ (aq) (c) 3 Fe2+ (aq) Fe(s) + 2...
-
A bond has an expected return of 15.33%, sensitivity to the term factor of 1.95 and to the default factor of 0.65. The current term risk premium is 3.01%. The risk-free rate of return is 3%. What is...
-
In the production-sales cycle, what are the four points at which quality costs are incurred? How are these costs interrelated through these points?
-
Which of the following is not true regarding the SECs allegations against Adelphia? (a) The company concealed rampant selfdealing by the family that founded and controlled Adelphia. (b) The company...
-
The catalytic dehydrogenation of 1-butene \[ \mathrm{C}_{4} \mathrm{H}_{8}(\mathrm{~g}) ightarrow \mathrm{C}_{4} \mathrm{H}_{6}(\mathrm{~g})+\mathrm{H}_{2}(\mathrm{~g}) \] is carried out at \(900...
-
The cash account for Pala Medical Co. at June 30, 20Y1, indicated a balance of $146,035. The bank statement indicated a balance of $181,965 on June 30, 20Y1. Comparing the bank statement and the...
-
Assume Spindrift Beverage Co. manufactures two products, flavored water and carbonated water, both on the same assembly lines and packaged 30 cans per pack. The predicted sales are 150,000 packs of...
-
Worldwide production of silicon carbide, SiC, is several hundred thousand tons annually. If you want to produce 1.0 10 5 metric tons of SiC, what mass (metric tons) of silicon sand (SiO 2 ) will you...
-
(a) Heating barium oxide in pure oxygen gives barium peroxide. Write a balanced equation for this reaction. (b) Barium peroxide is an excellent oxidizing agent. Write a balanced equation for the...
-
Ethical behavior is fundamental to communications between investors and companies. However, it is difficult for company founders to control their enthusiasm in discussions related to their company,...
-
What is float?
-
Describe how seasonal volume fluctuations influence current asset levels and financing requirements.
-
What is the goal of current accounts management?
-
How might a hospital that expects to have a cash shortage sometime during the coming year make sure that needed funds will be available?
-
What are accruals, and what is their role in short-term financing?
-
What is a pass-through scheme and how does it differ from a typical shell company billing scheme?
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
In Section 5.4, we showed that the radius of a geosynchronous orbit about the Earth is 4.2 107 m, compared with the radius of the Earth, which is 6.4 106 m. By what factor is the force of gravity...
-
You are an astronaut (m = 95 kg) and travel to a planet that is the same radius and mass as the Earth, but it has a rotational period of only 2 h. What is your apparent weight at the equator of this...
-
Suppose the density of the Earth was somehow reduced from its actual value to 1000 kg/m 3 (the density of water). Find the value of g, the acceleration due to gravity, on this new planet. Assume the...
-
A 500 mm long rotating shaft with a solid circular cross section is supported at its ends by bearings that can be modeled as simple supports. The middle of the shaft has a notch with a 1.25 mm...
-
4. (25 points) In the system shown in Figure 2, the rod is massless and the displacement y of the mass m is measured from its equilibrium position. Use the energy method to obtain the equation of...
-
1) A round bar of a brass with 5 cm diameter is to be reduced to a rod 3.25 cm in diameter by drawing. Suggest a procedure to be followed if a final yield strength of 350 MPa (or greater) is to be...

Study smarter with the SolutionInn App


