Using the data in Table 20.2, estimate the minimum mass of solid that would remain after evaporation
Question:
Using the data in Table 20.2, estimate the minimum mass of solid that would remain after evaporation of 1.0 L of seawater.
Data given in Table 20.2
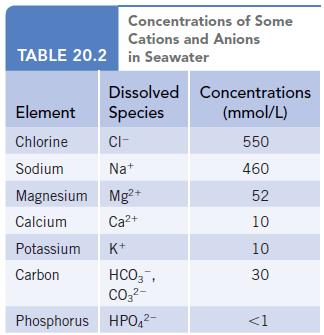
Transcribed Image Text:
Concentrations of Some Cations and Anions TABLE 20.2 in Seawater Dissolved Concentrations Species (mmol/L) 550 460 52 10 10 Element Chlorine CI- Sodium Na+ Magnesium Mg²+ Calcium Ca²+ Potassium K+ Carbon HCO3, CO3²- Phosphorus HPO4²- 30 <1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Dissolved Species Element Concentrations mmolL Chloride Cl 550 Sodium Na 460 Magnesium Mg2 52 Calciu...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The Tastee Bakery Company supplies a bakery product to many supermarkets in a metropolitan area. The company wishes to study the effect of the height of the shelf display employed by the supermarkets...
-
In Chapters 2 (Section 2.8) and 3 (Section 3.7) we analyzed the risk and return of the Orange County portfolio, using the building blocks of zero coupon bonds and duration in this exercise, we...
-
Using the data in Table 21.1, compare the price on July 24, 2009, of the following options on JetBlue stock to the price predicted by the Black-Scholes formula. Assume that the standard deviation of...
-
Evaluate the expression for the given value(s) of the variable(s). 4x - 7, x = 5/2
-
Ross-Simmons sued Weyerhaeuser for antitrust violation for driving it out of business. The claim was that Weyerhaeuser consistently outbid Ross for logs to process into lumber. Ross contended this...
-
In Markov analysis, the state probabilities must a. sum to 1. b. be less than 0. c. be less than 0.01. d. be greater than 1. e. be greater than 0.01.
-
Based on the following pedigree for a trait determined by a single gene (affected individuals are shown as filled symbols), state whether it would be possible for the trait to be inherited in each of...
-
Presented below is an aging schedule for Halleran Company. At December 31, 2014, the unadjusted balance in Allowance for Doubtful Accounts is a credit of $12,000. Instructions (a) Journalize and post...
-
Consider the region R on Earth including points whose coordinates, when rounded to a whole number of seconds, round to 1233'8" N, 8843'11" W. Determine with justification the perimeter of region R.
-
In any solution, there must be a balance between the positive and negative charges of the ions that are present. Use the data in Table 20.2 to determine how close to a balance of charge is achieved...
-
Which of the following statements is not correct? (a) Phosphates enhance the growth of algae in surface water. (b) Melting of the polar ice cap will result in a rise in sea levels. (c) Heavy metal...
-
Show that the matrix given by the cubic spline basis functions is positive definite.
-
A project is expected to sell one million units initially at a price per unit of $16.50 and a cost per unit of $6.50. Units sold will increase by 10% each year over the project's 3 year life....
-
Discussion will focus on understanding the selection of the direction of intervention with the client and how an activity analysis is crucial to the practice of occupational therapy. Discussion...
-
You are supporting an individual at the grocery store. You both get to the cash register to check out and realize that they do not have enough money to purchase all the items. The individual becomes...
-
The required rate by the bank is equal to ? Would the bank accept Mrs. Thompson's request? Mrs. Thompson asks her bank for a personal loan to be contracted for a 1-year period at an interest rate of...
-
If inflation is anticipated to be 1 percent during the next year, while the nominal rate of interest is 4 percent, what is the real interest rate for one year risk free loan?
-
This problem is useful for testing the ability of financial calculators and computer software. Consider the following cash flows. How many different IRRs are there? Year Cash Flow 0 ......... -$...
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
If the electric field vector E(z, t) of an EM wave in vacuum is, at a certain location and time, given by vector E = (10 V/m)(cos 0.5) i write an expression for the associated vector B-field.
-
The E-field of an electromagnetic wave is described by Write an expression for the B-field. Determine vector B(0, 0). 3 (i + jEo sin (kz ot + /6)
-
Starting with Eq. (3.32), prove that the energy densities of the electric and magnet fields are equal (u E = u B ) for an electromagnetic wave. (3.32) UB 2o
-
The negative news caused WACC to go up how much? Stock's beta 1.67 Market Risk Premium 4.1% Current Yield To Maturity on Treasury Bonds 3.1% Percent of Target Capital Structure in Debt 37% Spread on...
-
You work for Apple. After toiling away on $9.3 million worth of prototypes, you have finally produced your answer to Google Glasses: iGlasses (the name alone is genius). iGlasses will instantly...
-
What is the internal growth rate of Taylor, Inc. if the dividend payout ratio remains constant? Taylor, Inc. 2015 Income Statement Net Sales $28,900 Less: Cost of Goods Sold 23,400 Less: Depreciation...

Study smarter with the SolutionInn App


