What is the equilibrium constant for the conversion of -D-glucose to -D-glucose in water at room temperature
Question:
What is the equilibrium constant for the conversion of α-D-glucose to β-D-glucose in water at room temperature (assume that there is no glucose present in the open-chain structure)?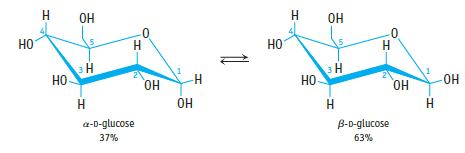
Transcribed Image Text:
НО H НО ОН m Т H H a-D-glucose 37% OH ОН H НО т НО ОН 10 3H Н H B-D-glucose 63% ОН H -ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The equilibrium constant for the conversion of Dglucose to Dglucose in water at room temper...View the full answer

Answered By

Jehal Shah
I believe everyone should try to be strong at logic and have good reading habit. Because If you possess these two skills, no matter what difficult situation is, you will definitely find a perfect solution out of it. While logical ability gives you to understand complex problems and concepts quite easily, reading habit gives you an open mind and holistic approach to see much bigger picture.
So guys, I always try to explain any concept keeping these two points in my mind. So that you will never forget any more importantly get bored.
Last but not the least, I am finance enthusiast. Big fan of Warren buffet for long term focus investing approach. On the same side derivatives is the segment I possess expertise.
If you have any finacne related doubt, do reach me out.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
What is the equilibrium constant for the following reaction at 25°C? Mg(s) + Zn2+ (aq)--Mg2+ (aq) + Zn(s)
-
The reaction SO2 1g2 + 2 H2S(g) 3 S(s) + 2 H2O (g) is the basis of a suggested method for removal of SO2 from power-plant stack gases. The standard free energy of each substance is given in Appendix...
-
My division had another great year last year. We all worked hard, and the results were there. But again we got no reward for our hard work. It's very frustrating. - Division Manager, General Products...
-
The cost of energy consumed in producing good units in the Baking Department was $150,000 and $154,000 for October and November, respectively. The number of equivalent units produced in October and...
-
If XYZ goes ahead and secures the backdated order and recognizes it as a bill and hold sale, which of the following is correct? (a) This would cause operating income to exceed CFFO by \($10,000\. (b)...
-
Consider a recent health-related situation you were involved in. Which life situation (Sect. 1.2) does it correspond to and what was your role in this life situation (Sect. 1.3)? List some of the...
-
Your firm is planning to issue preferred stock. The stock is expected to sell for $98 a share and will have a $100 par value on which the firm will pay a 14 percent dividend. What is the cost of...
-
A battery of 12 V is connected across capacitors of capacitances C1-C2=C3 C4 =C7= B F, Cs = C6= F. 12 V C3 C2 C1 i. What is the equivalent capacitance Ceq of the capacitors, you should show your work...
-
Draw two Lewis structures for the dipeptide alanineisoleucine that show the resonance structures of the amide linkage.
-
The body processes ethanol by first converting it to acetaldehyde in a reaction catalyzed by the enzyme alcohol dehydrogenase. In the body, acetaldehyde can cause headaches and nausea and is one of...
-
Construct the indicated confidence intervals for (a) The population variance 2 and (b) The population standard deviation . Assume the sample is from a normally distributed population. c = 0.99, s 2...
-
What is the difference between front-office Information systems and back-office information systems?
-
What are the differences between electronic commerce (e-commerce) and electronic business (e-business)?
-
Why are good interpersonal communication skills essential for system analysts?
-
What kind of knowledge and skills should a system analyst possess?
-
Why do we need to identify the information system architecture?
-
Potomac Automotive Co. manufactures engines that are made only on customers orders and to their specifications. During January, the company worked on Jobs 007, 008, 009, and 010. The following...
-
Consider the activities undertaken by a medical clinic in your area. Required 1. Do you consider a job order cost accounting system appropriate for the clinic? 2. Identify as many factors as possible...
-
For the circuit in Fig. 3.69 , find v 1 , v 2 , and v 3 using nodal analysis. 240 V (+- 201 V3 20 V2 1 40 40 10
-
For the circuit in Fig. 3.70 , find v 1 and v 2 using nodal analysis. 21. 20 V2 ww 10 30 V
-
Determine v 1 and v 2 in the circuit of Fig. 3.71 . 10 10 V1 V2 + v. 10 20 10 V (+ 5
-
Solve for x. 2+ In(x+3)=5 Do not round any intermediate computations, and round your answer to the nearest hundredth.
-
The fiscal year ends December 31 for Lake Hamilton Development. To provide funding for its Moonlight Bay project, LHD issued 7% bonds with a face amount of $600,000 on November 1, 2024. The bonds...
-
Your basal metabolic rate is the amount of energy (in calories) your body needs to function at rest. The Harris-Benedict formula is used to estimate the basal metabolic rate. There is one formula for...

Study smarter with the SolutionInn App


