When an electric current is passed through an aqueous solution of NaCl, the valuable industrial chemicals H
Question:
When an electric current is passed through an aqueous solution of NaCl, the valuable industrial chemicals H2(g), Cl2(g), and NaOH are produced. 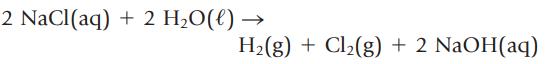
What mass of NaOH can be formed from 15.0 L of 0.35 M NaCl? What mass of chlorine is obtained?
Transcribed Image Text:
2 NaCl(aq) + 2 H₂O(l) - H₂(g) + Cl₂(g) + 2 NaOH(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To find the mass of NaOH formed and the mass of chlorine obtained you can use the given information ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
1) Emily wants to buy a computer that has a price of $1500, and she decides to pay for it with installments over 5 years. If the interest rate is 10 %, what is the monthly payment? 7) Suppose Crystal...
-
Indium(III) phosphide is a semiconducting material that has been frequently used in lasers, light-emitting diodes (LED), and fiberoptic devices. This material can be synthesized at 900. K according...
-
When an electric current is passed through molten sodium chloride, a. sodium metal is deposited at the positive electrode b. sodium ions are deposited at the positive electrode c. chlorine gas is...
-
What is the type of the expressions computed on these two lines? 4 > 5 print (4>5)
-
Describe a quality control chart and how it can be used. What are upper and lower control limits? What does it mean if an observation falls outside the control limits?
-
Astronomers view light coming from distant galaxies moving away from Earth at speeds greater than 10% of the speed of light. How fast does this light meet the telescopes of the astronomers?
-
For a certain two-dimensional flow field \[ \begin{aligned} & u=0 \\ & v=V \end{aligned} \] (a) What are the corresponding radial and tangential velocity components? (b) Determine the corresponding...
-
Three hundred gallons of a mixture containing 75.0 wt% ethanol (ethyl alcohol) and 25% water (mixture specific gravity = 0.877) and a quantity of a 40.0 wt% ethanol60% water mixture (SG = 0.952) are...
-
Sunspot Beverages, Limited, of Fiji uses the weighted-average method of process costing. It makes blended tropical fruit drinks in two stages. Fruit juices are extracted from fresh fruits and then...
-
Hydrazine, N 2 H 4 , a base like ammonia, can react with sulfuric acid. What mass of hydrazine reacts with 250. mL of 0.146 M H 2 SO 4 ? 2 NH(aq) + HSO4(aq) 2 NH5+ (aq) + SO4- (aq)
-
What mass of Na 2 CO 3 , in grams, is required for complete reaction with 50.0 mL of 0.125 M HNO 3 ? NaCO3(aq) + 2 HNO3(aq) 2 NaNO3(aq) + CO(g) + HO(l)
-
What is the difference between fluorescence and phosphorescence?
-
Saskatoon Dinner Company produces 'take - away' lunches, which it sells for $ 2 0 each. During the current month, the company produced 2 , 8 0 0 meals, but only sold 2 , 7 0 0 meals. The variable...
-
Griffin buys and sells ladies handbags. The average selling price is $ 4 1 0 per bag. Average variable costs ( purchase price of bags plus freight costs and selling expenses ) amounts to $ 1 9 0 per...
-
Saleena was hired and agreed to a $48.50 per hour compensation, for a regular 44 hour work week. Calculate her net earnings for the year. (10 marks) Federal Tax Brackets In Canada 2022 Federal Tax...
-
What quality assurance protocols do you employ to test and validate data integrity in software applications and databases ?
-
A company pays $ 5 , 0 0 0 for equipment. Annual depreciation on the equipment is $ 5 0 0 . What is the book value of the equipment at the end of Year 2 ?
-
Obtain the annual reports of as many companies as you have team membersone company per team member. Most companies post their financial statements on their websites. Requirements 1. Identify the...
-
6. (Potential Energy and Conservation of Energy) What should be the spring constant k of a spring designed to bring a 1200-kg car to rest from a speed of 95 km/h so that the occupants undergo a...
-
Propose a plausible synthesis for the following transformation. CH3 CH3 CH3
-
Calculate S for the isothermal compression of 1.75 mol of Cu(s) from 2.15 bar to 1250. bar at 298 K. = 0.492 10 4 K 1 , = 0.78 10 6 bar 1 , and the density is 8.92 g cm 3 . Repeat the calculation...
-
Calculate S for the reaction 3H 2 (g) + N 2 (g) 2NH 3 (g) at 725 K. Omit terms in the temperature-dependent heat capacities higher than T 2 /K 2 .
-
Pharoah, Inc. began work on a $6,909,000 contract in 2025 to construct an office building. During 2025, Pharoah, Inc. incurred costs of $1,791,800, billed its customers for $1,174,000, and collected...
-
On January 1, 2016, Poplar Company acquired 80% of the common stock of Sequoia Company for $400,000. On this date, Sequoia had total owners' equity of $400,000. The excess of cost over book value was...
-
Frank gave Betty real estate with a basis of $100,000 and marketable securities with a basis of $20,000. In exchange, Betty gave Frank real estate with a basis of $240,000 and worth $300,000. The...

Study smarter with the SolutionInn App


