Write the net ionic equation and identify the spectator ion or ions in the reaction of nitric
Question:
Write the net ionic equation and identify the spectator ion or ions in the reaction of nitric acid and magnesium hydroxide. What type of reaction is this?
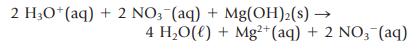
Transcribed Image Text:
2 H3O+ (aq) + 2 NO3(aq) + Mg(OH)₂(s) → 4 H₂O(l) + Mg²+ (aq) + 2 NO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The given reaction is a neutralization reaction between nitric acid H3O and magnesium hydroxide MgOH...View the full answer

Answered By

Diksha Bhasin
I have been taking online teaching classes from past 5 years, i.e.2013-2019 for students from classes 1st-10th. I also take online and home tuitions for classes 11th and 12th for subjects – Business Studies and Economics from past 3 years, i.e. from 2016-2019. I am eligible for tutoring Commerce graduates and post graduates. I am a responsible for staying in contact with my students and maintaining a high passing rate.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
Write balanced net ionic equations for the reactions that occur in each of the following cases. Identify the spectator ion or ions in each reaction. (a) Cr2 (SO4)3(aq) + (NH4)2CO3(aq) (b) Ba...
-
The mineral dolomite contains magnesium carbonate. This reacts with hydrochloric acid. (a) Write the net ionic equation for this reaction and identify the spectator ions. (b) What type of reaction is...
-
What is the is the effect of abolition of an import quota on (i) national saving, (ii) domestic investment, (iii) NCO, (iv) the real exchange rate, and (v) net exports?
-
Other things equal, how would you expect the following shifts to affect a currencys real exchange rate against foreign currencies? a. The overall level of spending doesnt change, but domestic...
-
From the U.S. Census Bureau and the article "U.S. Prison Population Falls for Third Year" by B. Montopoli of CBS News, we found that about 1 in every 200 Americans are in prison. Use this information...
-
A six cylinder four stroke I.C. engine is to develop \(100 \mathrm{~kW}\) IP at \(800 \mathrm{rpm}\). The stroke to bore ratio is 1.25 . Assuming mechanical efficiency of \(80 \%\) and brake mean...
-
Baker Brothers has a DSO of 40 days, and its annual sales are $7,300,000. What is its accounts receivable balance? Assume that it uses a 365-day year.
-
Lets say a portion of money that you add, usually, in a package at Quellenhof to cover the fee. Example: a hotel room costs 200 and the flight 100. Total 300. However, the guest did not book it...
-
Name two anions that combine with Al 3+ ion to produce water-soluble compounds.
-
Give the formula for each of the following compounds: (a) A soluble compound containing the acetate ion (b) An insoluble sulfide (c) A soluble hydroxide (d) An insoluble chloride (e) A strong Brnsted...
-
The most important job of the Federal Trade Commission today is to _______. a) prevent false and deceptive advertising b) break up unlawful trusts c) issue cease-and-desist orders when...
-
Can you elaborate on the role of ethical leaders in promoting social responsibility and sustainability initiatives within organizations, and how do they balance the pursuit of profit with broader...
-
Van Duzer explains that one of the consequences of the Fall on the nature of work is that people are working harder at a faster pace, not being able to take a pause, under the influence of increasing...
-
1. What is tension? 2. What does tension do to materials? 3. What is compression? 4. What does compression do to materials?
-
You decide to accept a quote from a store specializing in office remodeling. They offer to pay in quarterly installments due over 3 years, giving a footing of $600,000. of $600,000. The estimate as...
-
Corporate) taxes play an important role in making investment decisions. Taxes influence the incremental cash flows needed to assess the feasibility of an investment. Explain why and how taxes are...
-
On September 1, 2011, Parcel Corporation purchased 80% of the outstanding common stock of Sack Corporation for $152,000. On that date, Sacks net book values equaled fair values, and there was no...
-
Repeat Exercise 16.6 using the t-test of the coefficient of correlation. Is this result identical to the one you produced in Exercise 16.6?
-
In an adiabatic compression of one mol of an ideal gas with C V ,m = 5/2 R, the temperature rises from 278 K to 450. K. Calculate q, w, H, and U.
-
Can the following compound be prepared via a Williamson ether synthesis? Explain your answer.
-
Calculate H o R and U o R for the oxidation of benzene (g). Also calculate -U R.
-
Research the civil case Katz, et al. v. Panera Bread Co. and answer the following questions: Give a brief description of the lawsuit. Did Panera properly display the contents of the drink? Explain...
-
Waiting in the South Bend, Indiana jail for the trial on the properties that nana (grandma) purchased in New York for the trial that was upcoming and to be held in the Northern District of Indiana....
-
Should a defendant be forced to take medication in order to be competent to stand trial. A 2008 review of the twelve largest states indicated that the number of accused felons that were declared by...

Study smarter with the SolutionInn App


