Azomethane, CH 3 NNCH 3 , is not a stable compound, and once generated, it decomposes. The
Question:
Azomethane, CH3NNCH3, is not a stable compound, and once generated, it decomposes. The rate of decomposition was measured by monitoring the partial pressure of azomethane, in torr:
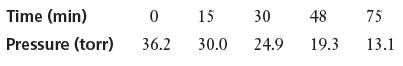
Plot the data and determine the instantaneous rate of decomposition of azomethane at t = 20 min.
Transcribed Image Text:
Time (min) Pressure (torr) 0 36.2 15 30 30.0 24.9 48 19.3 75 13.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Estimating th...View the full answer

Answered By

Keziah Thiga
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagirism), well-researched and critically analyzed papers.
4.90+
1504+ Reviews
2898+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
There are two significant pieces of legislation that impact maritime security in the United States. These include the Maritime Transportation Security Act 2002 and the S.A.F.E. Port Act of 2006....
-
At 400 K, the rate of decomposition of a gaseous compound initially at a pressure of 12.6 kPa was 9.71 Pa S-1 when 10.0 per cent had reacted and 7.67 Pa S-I when 20.0 per cent had reacted. Determine...
-
The rate of the decomposition of hydrogen peroxide, H 2 O 2 , depends on the concentration of iodide ion present. The rate of decomposition was measured at constant temperature and pressure for...
-
(a) Find the Maclaurin series for the function f(x)= ln(1+x) and hence that for In(1+x) (b) By keeping the first four terms in the Maclaurin series for In(1+x) integrate the function In(1+x) from x =...
-
Management for Marlowe Manufacturing Company decided in 2010 to discontinue one of its unsuccessful product lines. (The product line does not meet the definition of a business component.) The planned...
-
Use all three methods in this Section to find solutions to within 107 for the following problems. a. x2 4x + 4 ln x = 0 for 1 x 2 and for 2 x 4 b. x + 1 2 sin x = 0 for 0 x 1/2 and for 1/2 ...
-
Dating and disclosure. Refer to the Journal of Adolescence (Apr. 2010) study of adolescents disclosure of their dating and romantic relationships, Exercise 8.43 (p. 419). Data collected for a sample...
-
Ray and Maria Gomez have been married 3 years. They live at 1610 Quince Ave., McAllen, TX 78701. Ray works for Palm Oil Corporation and Maria works for the City of McAllen. Maria's Social Security...
-
\ table [ [ Equipment , , ] , [ Jan . 1 Bal., 8 0 , 0 0 0 , Nov. 1 0 Cost of equipment sold ] , [ July 3 1 Purchase of equipment, 3 5 , 0 0 0 , ] , [ Sept . 2 Purchase of equipment, 2 6 , 5 0 0 , ] ,...
-
In a method of initial rates experiment, is the measured rate an average rate or an instantaneous rate? Explain.
-
A gas, AB, decomposes and the volume of B 2 produced is measured as a function of time. The data obtained are as follows: What is the average rate of production of B 2 for the first 8.3 min? For the...
-
An article in the Wall Street Journal gave the following explanation of how products were traditionally priced at Parker-Hannifin Corporation: For as long as anyone at the 89-year-old company could...
-
In the following circuit, the supply voltage is 12V. Suppose VIN = +7 V, the output voltage will be, VIN VOUT R1 10 R2 10
-
Find the derivative of the function f(x)=(7x-8)". Find the derivative of the function y=- 2x x+6 Find the derivative of the function y = In Find the derivative of the function f(t): Find the...
-
Use the information below to answer questions 29 - 39. Make a table similar to examples 12 and 13 to help answer the questions. Ten persons of students at ACM are nursing students. Forty percent of...
-
Abby Industries, Inc. has the following capital structure. Type Amount Rate of Return Mortgages (debt) $25,000,000 7% Bonds (debt) 180,000,000 9% Common stock (equity) 100,000,000 10% Preferred stock...
-
Review the network of stakeholders Choose five different stakeholders and provide examples of why a project manager would need to negotiate with that stakeholder. FIGURE 10.1 Network of Stakeholders...
-
Draw all possible products of the chlorination of (S)-2-bromo-l,l-dimethylcyclobutane. Specify whether they are chiral or achiral, whether they are formed in equal or unequal amounts, and which are...
-
Repeat the previous problem, but close the positions on September 20. Use the spreadsheet to find the profits for the possible stock prices on September 20. Generate a graph and use it to identify...
-
Identify the reagents necessary to make each of the following amino acids via the amidomalonate synthesis. (a) Isoleucine (b) Alanine (c) Valine
-
Each of the following carboxylic acids was treated with bromine and PBr3 followed by water, and the resulting α-haloacid was then treated with excess ammonia. In each case, draw and...
-
Each of the following aldehydes was converted into an -amino nitrile followed by hydrolysis to yield an amino acid. In each case, draw and name the amino acid that was produced. (a) Acetaldehyde (b)...
-
thumbs up if correct A stock paying no dividends is priced at $154. Over the next 3-months you expect the stock torpeither be up 10% or down 10%. The risk-free rate is 1% per annum compounded...
-
Question 17 2 pts Activities between affiliated entities, such as a company and its management, must be disclosed in the financial statements of a corporation as O significant relationships O segment...
-
Marchetti Company, a U.S.-based importer of wines and spirits, placed an order with a French supplier for 1,000 cases of wine at a price of 200 euros per case. The total purchase price is 200,000...

Study smarter with the SolutionInn App


