Consider a sample of an ideal gas with n and T held constant. Which of the graphs
Question:
Consider a sample of an ideal gas with n and T held constant. Which of the graphs below represents the proper relationship between P and V? How would the graph differ for a sample with a larger number of moles?
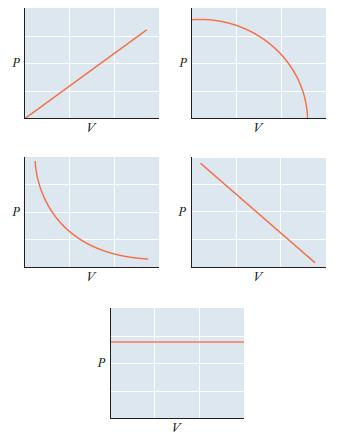
Transcribed Image Text:
P P V V P P P V V V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The graph on the left represents the proper relationship between P and V for a sample of an ideal ga...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
(1 point) Write the system of equations as a matrix equation, that is, rewrite it in the form Input your answer below: a11 a12 a13 = a21 = a22= = a23 = a31 b3 = a32 = a33 = b = = b2 = || || - -2x +...
-
Consider 2.00 moles of an ideal gas that is taken from state A (PA = 2.00 atm, VA = 10.0 L) to state B (PB = 1.00 atm, VB = 30.0 L) by two different pathways. These pathways are summarized in the...
-
In 1858, J. Waterston found a clever way to estimate molecular sizes from macro- scopic properties of a liquid, by comparing its surface tension and heat of vaporiza- tion. The surface tension of...
-
A firm has total debt of $6,000,000 and stockholder's equity is $4,000,000. The firm wants to calculate equity-to- total asset ratio in order to make decision about further raise of capital. What is...
-
Palmer Corporation operates on a calendar-year basis. It begins the annual budgeting process in late August when the president establishes targets for the total dollar sales and net income before...
-
The head of the information systems group at Conway Consulting must assign six new analysts to offices. The following closeness matrix shows the expected frequency of contact between analysts. The...
-
Identify the six different discovery devices. What is the purpose of each device?
-
Briefly discuss the components of e-manufacturing. In what ways can companies collaborate in producing goods and services? Report on at least one Web source on collaborative manufacturing or...
-
Calculate the value of the property based on the information provided below. Name Nuhaw Wahl Project Address 321 Station Street, Canterbury, NSW Net operating income $953,000 Operating expenses (per...
-
Consider a sample of N 2 gas under conditions in which it obeys the ideal gas law exactly. Which of these statements is true? (a) A sample of Ne(g) under the same conditions must obey the ideal gas...
-
Why do heavier gases move more slowly than light gases at the same temperature?
-
Tru Developers, Inc., sells plots of land for industrial development. Tru recognizes income for financial reporting purposes in the year it sells the plots. For some of the plots sold this year, Tru...
-
82. The Near Earth Asteroid Rendezvous (NEAR) spacecraft, after traveling 2.2 billion km, is meant to orbit the asteroid Eros with an orbital radius of about 25 km. Eros is roughly 40 km X 6 km X 6...
-
Morganton Company makes one product, and has provided the following information to help prepare the master budget for its first four months of operations: The budgeted selling price per unit is $60....
-
A test rocket is fired vertically upward from a well. A catapult gives it initial speed 50 m/s at ground level. Its engines then fire and it accelerates upward at 5.0 m/s 2 until it reaches an...
-
Gabbe Industries is a division of a major corporation. Last year the division had total sales of $24,049,700, net operating income of $4,641,592, and average operating assets of $8,293,000. The...
-
On 2 October 20X4, a national hardware retailer, One Hardware Corporation, announced a formal plan to refocus its operations. As part of the plan, management decided to sell its portfolio of...
-
Why is there a different XBRL taxonomy for each country, including XBRL Australia, XBRL Canada, XBRL Germany, XBRL Japan, XBRL-Netherlands, XBRL-US, and XBRL UK? What would happen if there were only...
-
Using the parallel-axis theorem, determine the product of inertia of the area shown with respect to the centroidal x and y axes. 6 in. 9 in. 9 in- 4.5 in. in. 4.5 in.
-
An ideal gas sample containing 1.75 moles for which C V ,m = 5/2R undergoes the following reversible cyclical process from an initial state characterized by T = 275 K and P = 1.00 bar: a. It is...
-
For protein denaturation, the excess entropy of denaturation is defined as is the transition excess heat capacity. The way in which δC trs P can be extracted from differential scanning...
-
The standard entropy of Pb(s) at 298.15 K is 64.80 J K -1 mol - 1 . Assume that the heat capacity of Pb(s) is given by The melting point is 327.4C and the heat of fusion under these conditions is...
-
LG just paid a $2 annual dividend on its common stock. The dividend is expected to increase 5.5% per year indefinitely. If the required rate of return is 7.8%, What is the the stock's value next year?
-
Suppose Peter retires at age 60 with $1,000,000 in pension. How much will he and his beneficiaries receive if he requests 3 equal periodic payments with the first one to be received at age 60 and the...
-
1. Determine the stability condition for the given structure assuming that the links are rigid. (sina sino -2lcosa -2lcose- k www ||

Study smarter with the SolutionInn App


