Consider the Lewis structure below. Is this an ion? If so, what is its charge? == :O:
Question:
Consider the Lewis structure below. Is this an ion? If so, what is its charge?
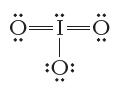
Transcribed Image Text:
ة==ة :O:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
No the Lewis structure you sent is not an ion It is a molecule of ozone O3 An ion is an atom or mole...View the full answer

Answered By

Amit Kumar
I am a student at IIT Kanpur , which is one of the prestigious colleges in INDIA.
Cleared JEE Advance in 2017.I am a flexible teacher because I understand that all students learn in different ways and at different paces. When teaching, I make sure that every student has a grasp of the subject before moving on.
I will help student to get the basic understanding clear. I believe friendly behavior with student can help both the student and the teacher.
I love science and my students do the same.
4.90+
44+ Reviews
166+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Consider the Lewis structure for glycine, the simplest amino acid: (a) What are the approximate bond angles about each of the two carbon atoms, and what are the hybridizations of the orbitals on each...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Briefly describe some common information system controls that need to be implemented by business managers, not IS professionals.
-
rn rn Anushka (an Australian Tax resident) works as an employee for a child care centre, Brilliant Kids Pty Ltd, on a permanent part-time basis while she runs her business as a day carer for her own...
-
What is a real option? What are some types of real options? Shrieves Casting Company is considering adding a new line to its product mix, and the capital budgeting analysis is being conducted by...
-
In Problem S10-32 Phillups Corporation is just as concerned that its U.S. distributors receive shipments in the minimum amount of time as they are about minimizing their shipping costs. Suppose each...
-
What do you like best about working at this company?
-
Polk Company manufactures basketballs. Materials are added at the beginning of the production process and conversion costs are incurred uniformly. Production and cost data for the month of July 2017...
-
How to create a relational model from DDL statements and how you save a relational model design.?
-
The following molecules have similar formulas, but each has a different shape. Draw Lewis structures for these compounds and then determine their molecular shapes. (a) SiF 4 , (b) KrF 4 , (c) SeF 4
-
The N 5 + cation has been synthesized and studied. Consider the possible Lewis structure below. Indicate the hybridization expected for each nitrogen atom and the expected bond angles. Assuming that...
-
Karen belongs to a number of registered pension schemes. Until tax year 2009 -10 she had received no benefits from any of these schemes but in October 2009 she took a lump sum of 350,000 from one of...
-
Write out the expression for put-call parity in terms of the Bond price. Explain the characteristics of the bond: maturity, coupon and face value.
-
Describe the pre-incident planning process utilized by the organization you represent. o If you are not currently affiliated with an organization, select an organization of your choice. Conduct...
-
Consider a transportation corporation named C.R. Evans, which has just completed the development of a new subway system in a medium-sized city in Western Canada. Currently, there are plenty of seats...
-
A middle school has 850 students. 255 students are in seventh grade. What percent of students are in seventh grade?
-
Jamaal rolls a six-sided die and spins a four-color spinner. What is the probability that he rolls a 4 and spins yellow?
-
Listed below are several terms and phrases associated with accounting for income taxes. Pair each item from List A with the item from List B (by letter) that is most appropriately associated withit....
-
Evaluate the integral, if it exists. Jo y(y + 1) dy
-
List the allowed quantum numbers m l and m s for the following subshells and determine the maximum occupancy of the subshells: a. 2p b. 3d c. 4 f d. 5g
-
The effective path length that an electron travels before being ejected into the vacuum is related to the depth below the surface at which it is generated and the exit angle by d = cos , where is...
-
The inelastic mean free path of electrons in a solid, , governs the surface sensitivity of techniques such as AES and XPS. The electrons generated below the surface must make their way to the surface...
-
Write the advantages and disadvantages your organization gained from internal and external sources of recruitment.
-
What are the methodological approaches used by symbolic interactionists in conducting research, such as participant observation, ethnography, and qualitative interviews, to explore the complexities...
-
The volume of the tank is 12L and the time elapsed is non-negative. For which values of t does the equation V=4t accurately model the volume of the tank?

Study smarter with the SolutionInn App


