Gallium metal has a melting point of 29.8C. Use the information below to estimate the boiling point
Question:
Gallium metal has a melting point of 29.8ºC. Use the information below to estimate the boiling point of gallium in °C.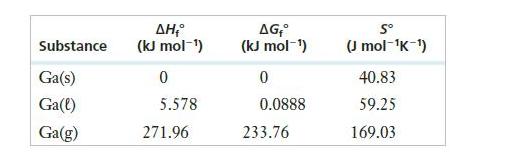

Transcribed Image Text:
Substance Ga(s) Ga(l) Ga(g) ΔΗ,° (kJ mol-¹) 0 5.578 271.96 AG, (kJ mol-¹) 0 0.0888 233.76 Sº (J mol-¹K-¹) 40.83 59.25 169.03
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
T ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
The element gallium has a melting point of 29.8C. If you hold a sample of gallium in your hand, should it melt? Explain briefly. Gallium metal Cengage Leaming/Charles D. Winters
-
The following picture represents atoms of hypothetical, nonmetallic, monatomic elements A, B, and C in a container at a temperature of 4 K (the piston maintains the pressure at 1 atm). None of these...
-
Find an article published in any newspaper, magazine, or credible online resource in the last two months regarding the balance of power between the state and Federal government. The article can...
-
In subsurface pipe drainage, describe the different types and materials used for the drainage?
-
What argument is given for reporting noncurrent operating assets at their historical costs instead of at current values?
-
For the last 2 years, The Health Company has experienced a fixed cost of $850,000 per year and an (r v) value of $1.25 per unit for its multivitamin line of products. International competition has...
-
Jerome M. Eisenberg is an antiquities dealer and a self-proclaimed expert in classical antiquities with a doctorate in Roman, Egyptian, and Near Eastern art. Maurice E. Hall Jr. is an art dealer who...
-
Georgia Pacific, a manufacturer, incurs the following costs. (1) Classify each cost as either a product or a period cost. If a product cost, identify it as a prime and/or conversion cost. (2)...
-
On June 30, 2016, Flint Limited issued 13.75% bonds with a par value of $802,000 due in 20 years. They were issued at 99 and were callable at 102 at any date after June 30, 2023. Because of lower...
-
The enthalpy of vaporization for water is 40.65 kJ mol -1 . As a design engineer for a project in a desert climate, you are exploring the option of using evaporative cooling. (a) If the air has an...
-
The molecular scale pictures below show snapshots of a strong acid at three different instants after it is added to water. Place the three pictures in the correct order so that they show the progress...
-
Use the superposition principle to find \(v_{\mathrm{O}}\) in Figure P \(_{3}=\) 32 . 15 V | 200 VO | 200 200 +1) 5V
-
Enhanced Interrogation Techniques. Should they be utilized? At a minimum, the paper will follow the following format. The student is encouraged to be creative with the research project. The format...
-
We would like to carry a metal piece of length L (ignore the thickness) in horizontal position around a corner from a corridor of 2 meters wide into a corridor of 3 meters wide as shown in the figure...
-
Sahari Aswad is running its business from 2001 in sultanate of Oman. It has cash in hand RO 40000, debtors RO 20000, stock in hand RO 10000, creditors RO 15000, capital RO 60000. The current ratio...
-
Description There is no need to include the caption for the petition. Draft the body of the petition using the 3 counts we discuss in class. Be sure to include all body sections and appropriate...
-
In what ways does James Joyce's Ulysses challenge traditional narrative structure and what effect does this have on the reader's experience?
-
Douglas Corporation had 120,000 ordinary shares outstanding on January 1, 2010. On May 1, 2010, Douglas issued 60,000 ordinary shares. On July 1, Douglas purchased 10,000 treasury shares, which were...
-
Data on weekday exercise time for 20 females, consistent with summary quantities given in the paper An Ecological Momentary Assessment of the Physical Activity and Sedentary Behaviour Patterns of...
-
The measured molar heat capacities for crystalline KCl are as follows at the indicated temperatures: a. Explain why the high-temperature limit for C V is apparently twofold greater than that...
-
The speed of sound is given by the relationship where C P is the constant pressure heat capacity (equal to C V + R), R is the ideal gas constant, T is temperature, and M is molar mass. a. What is the...
-
Consider the following energy levels and associated degeneracies for atomic Fe: a. Determine the electronic contribution to C V for atomic Fe at 150. K assuming that only the first two levels...
-
Identify a product / brand that is highly advertised. Research and analyze all the media that the company is using to promote the product. You need to answer the following: 1. About the product 2....
-
1. A. Discuss why organizations need to experience change?? Explain using scientific proofs. B. Evaluate the following situations in terms of change process and change types. Explain using scientific...
-
(a) Differentiate between the within and between estimators in panel data analysis. (5) (b) Explain the implications of not considering the correlation structure in panel data and how such a...

Study smarter with the SolutionInn App


